
Most people who get burned by a bad peptide vendor had no idea the product was problematic. They saw a professional-looking website, read the five-star reviews, checked for a certificate of analysis, and paid. What they didn't know was that those signals can all be faked.
A 2025 analysis of over 550 peptide suppliers found roughly 40% displayed at least one significant red flag. This guide gives you the checklist to catch those flags before they cost you money, or your health.
Start here: what kind of peptide are you actually buying?
The checklist looks completely different depending on your answer to this question.
Research peptides carry a "for laboratory use only, not for human consumption" label. That label is a legal shield for the seller, not a quality guarantee for you. Vendors who sell research peptides operate with no regulatory manufacturing oversight, no required testing standards, and no accountability for what's actually in the vial. Independent analysis has found roughly 25% of unregulated peptide products are mislabeled: the wrong compound, the wrong concentration, or both.
Pharmaceutical-grade peptides are medications. Either FDA-approved drugs, or compounds prepared by licensed compounding pharmacies operating under USP Chapter <797> sterile manufacturing standards, independent lot testing, and physician prescription requirements.
If you're shopping for research peptides, your job is to find vendors who voluntarily do what regulated pharmacies are legally required to do: batch-specific COA documentation, named testing labs, traceable manufacturing. That accountability is voluntary, meaning some vendors do it, most don't, and none are required to. If you're working with a telehealth provider or compounding pharmacy, the regulatory framework does most of the heavy lifting. Your job there is to verify the provider actually uses that framework, rather than just invoking it as marketing.
Why source matters as much as which peptide you choose goes deeper on this divide if you want the full picture before going further.
Red flags in online peptide vendors
A 2025 analysis of over 550 peptide suppliers found roughly 40% displayed at least one significant red flag. The signals are often subtle — professional websites, five-star reviews, and certificates of analysis that look legitimate but aren't.
These are the patterns that reliably indicate a problematic operation. Not every bad vendor checks all of these, and some bad vendors check none of them, but the more boxes a vendor hits, the harder it should be to justify buying from them.
No verifiable physical address or contact information
Legitimate businesses exist somewhere. If the only contact option is a web form or a generic customer service email, and you can't locate an actual address, phone number, or business registration, the seller has no accountability built in. Anything you buy is essentially anonymous.
Payment methods with no recourse
Vendors who only accept cryptocurrency or wire transfer are structuring a transaction you can't dispute. A credit card gives you a chargeback option if something goes wrong. Crypto and wire transfers do not. When a peptide business refuses credit card payments, there's a reason.
Prices dramatically below market rate
Pharmaceutical-grade peptide manufacturing costs real money: independent lab testing, sterile facilities, documented lot numbers, regulated cold chain logistics. A vial priced 70% below what a licensed compounding pharmacy charges is different somehow. The cost had to come from somewhere: cheaper sourcing, skipped testing, incorrect concentration, or all three.
Generic or copy-pasted product descriptions
Vendors who understand what they're selling describe it specifically. Template product descriptions duplicated across dozens of compounds suggest someone filled out a website without knowing (or caring) much about the actual chemistry. That's not who you want handling synthesis quality control.
Fabricated business history
You can check any domain's registration date at a free WHOIS lookup service. When a vendor claims "serving researchers since 2009" on a domain registered in 2024, that's a documented pattern in peptide vendor exit scams: fabricate a history to build false trust, disappear after complaints accumulate.
Injectables shipped without a prescription
In the US, shipping injectable peptides without a physician's prescription is operating outside the law: not a technicality, an actual enforcement target. A vendor who ignores this is telling you how they approach compliance more broadly.
Therapeutic marketing on "research-only" products
When a vendor sells vials labeled "not for human consumption" while simultaneously publishing dosing guides for human use, before-and-after transformation photos, and recovery protocols, they are marketing for a purpose they won't legally claim. The "research only" label is their liability shield. The marketing is what they actually sell.
How to verify a COA, and why most people don't
The Certificate of Analysis is the main document vendors use to demonstrate quality. It's also the most commonly fabricated document in the industry.
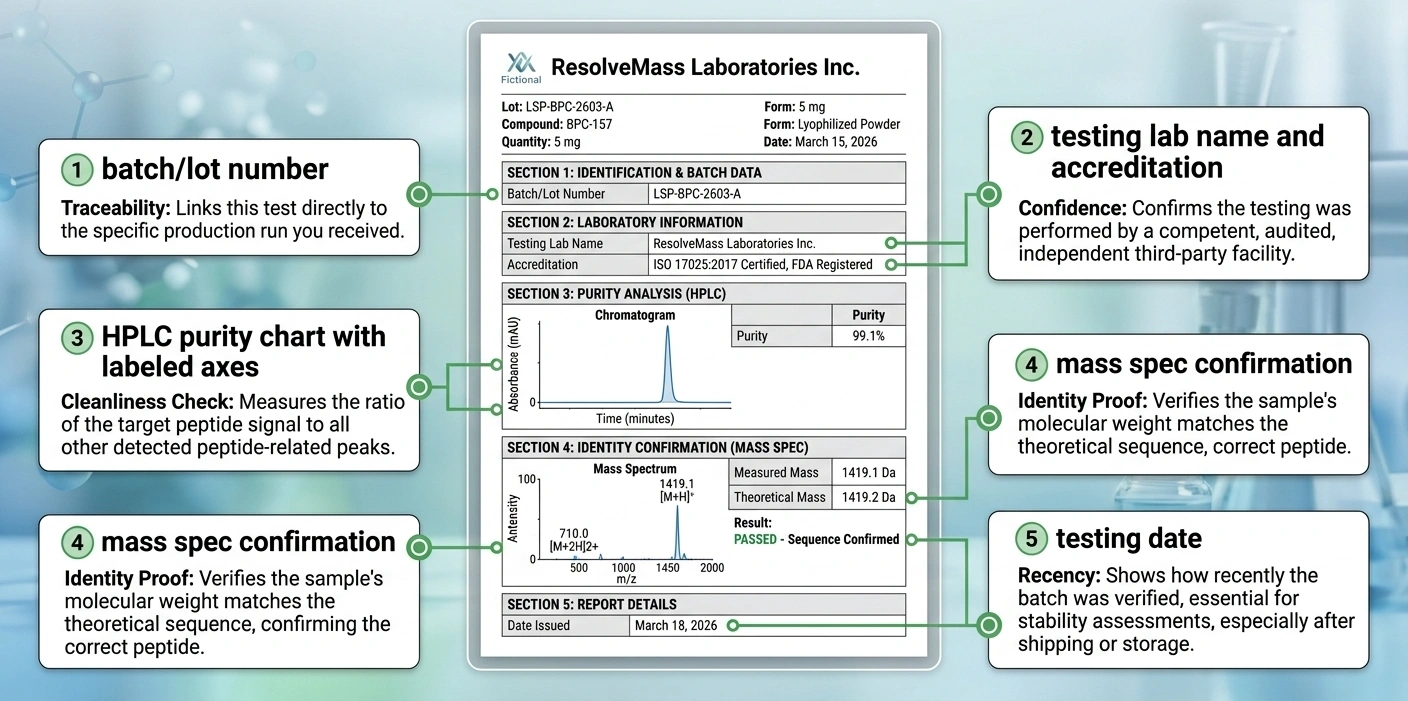
Here's what a real, usable COA contains:
A batch or lot number that matches the specific product you're buying. Not a generic document that applies to all inventory. The batch number on the COA should match the lot number on your order, and vendors should be able to provide this before you pay.
The testing lab's name, accreditation, and contact details. A real laboratory is a real business with a website, phone number, and staff who respond to email. You should be able to look them up.
HPLC purity analysis showing the purity percentage and the methodology used. Liquid chromatography-high resolution mass spectrometry (LCHRMS) detects more impurities than standard HPLC-UV methods. If a vendor provides LCHRMS data, that's a stronger signal than UV-only. If the COA contains only a purity number with no supporting methodology, that number is unverified.
Mass spectrometry confirming molecular identity. HPLC tells you how pure something is. Mass spec tells you what it actually is. Both together is meaningful quality documentation. One without the other is incomplete.
Actual data, not just conclusions. Charts with labeled axes, peak identification, methodology described. A COA that says "purity: 99.1%" with nothing behind that number is not verification.
A recent testing date. A COA from 2022 applied to inventory you're buying in 2026 doesn't tell you what's in your current batch. Batch-specific and recent is what accountability looks like.
The most important step most people skip: contact the lab directly. Legitimate third-party testing labs — Janoshik, MZ Biolabs, Colmaric Analyticals — are real companies that respond to inquiries and can confirm whether a document is theirs. If the lab listed on a COA doesn't respond, doesn't exist under that name, or can't find the report in their records, the document is fabricated.
COA checklist: verify lot matching, lab identity, analytical method, and testing recency.
What legitimate telehealth providers actually look like
If you want pharmaceutical-grade peptides under physician supervision, a licensed telehealth provider is the correct channel for anything you plan to inject. Here's what separates real clinical providers from wellness businesses using "telehealth" as a marketing word:
- Physician oversight with verifiable credentials. Board-certified physicians who hold state medical licenses and, where applicable, DEA licenses. The supervising physician's name and credentials should be findable on the provider's site. You can verify board certification through the American Board of Medical Specialties and state medical board licensing through public lookup tools. If the provider is vague about medical supervision, or if it turns out to be a health coach or nurse practitioner operating beyond their scope, that's a meaningful problem.
- A named, licensed compounding pharmacy as the source. Legitimate providers source from 503A or 503B compounding pharmacies, ideally PCAB-accredited (the voluntary gold standard for compounding quality). They should be willing to name the pharmacy. If they won't say where your medication is compounded, or the answer turns out to be a research supplier rather than a licensed pharmacy, you are not getting pharmaceutical-grade product.
- A real medical evaluation before any prescription. Lab work, medical history review, contraindication screening, not a 5-minute intake form. Peptides interact with hormonal therapies, blood pressure medications, and diabetes drugs in ways that aren't always obvious. A provider who prescribes without evaluating you is not providing healthcare. They're providing retail with a prescription header.
- Transparent, complete pricing upfront. The full cost, covering the consultation, prescription, and pharmacy, should be stated before you commit. If pricing is unclear or if line items appear after you've signed up, ask for the complete breakdown before you pay anything.
- Honest claims about what's currently available. The July 2026 FDA advisory committee meetings may move peptides including BPC-157 and TB-500 onto the 503A bulk drug substances list, which would allow licensed compounding pharmacies to prepare them. Until that happens, any provider claiming to sell pharmacy-grade versions of compounds still on the FDA's Category 2 list is misrepresenting their sourcing. Understanding the current regulatory status of specific compounds, like BPC-157 or semaglutide, matters when you're evaluating whether a provider's claims are accurate.
Questions to ask before you commit
These apply to vendors and telehealth providers alike. Vague or hostile answers are answers.
- Where is this peptide manufactured or compounded? A specific, verifiable answer. Not a country or a general claim about quality.
- Can I see the batch-specific COA for the exact lot I'm receiving? Emphasis on "batch-specific." Ask for the lot number the COA corresponds to.
- Which lab performed the testing, and how do I verify their accreditation?
- What are your cold chain protocols for shipping? Peptides degrade outside refrigeration. A vendor who ships without cold packs or temperature-controlled packaging has a quality problem that no COA can fix.
- (For telehealth providers) Which compounding pharmacy prepares the medication, and is it PCAB-accredited?
- (For telehealth providers) What physician is supervising my prescription, and what are their board certifications?
- What is your return or refund policy for damaged or unsatisfactory product?
If you're on any prescription medications, add: what drug interactions have been assessed for this compound? A responsible provider will either answer directly or refer you to the prescribing physician. A provider who dismisses the question hasn't thought about it.
Why price is a misleading signal
Cheap peptides are a red flag. The manufacturing infrastructure for pharmaceutical-grade compounds costs real money: independent lab testing, sterile facilities, regulated processes, documented lot numbers. There's no shortcut to a lower price that doesn't remove something from that process.
But expensive peptides aren't automatically legitimate. Several telehealth providers charge premium prices while sourcing from the same gray-market channels they advertise against. Price reflects cost structure. It doesn't verify what those costs went toward.
The most useful thing price tells you is when something is probably wrong. A vial priced far below what comparable pharmacy-grade product costs means something about the product is different. That's worth knowing. But high price alone is not verification. It just means you paid more.
Certifications and credentials to look for
For compounding pharmacies and telehealth providers:
- State pharmacy board license: publicly verifiable through state licensing databases
- PCAB accreditation: voluntary, but the recognized standard for compounding quality assurance
- 503A or 503B facility designation: 503A pharmacies compound for individual patients with prescriptions; 503B outsourcing facilities operate under stricter FDA cGMP standards at higher volume
- Physician board certification: verifiable through the American Board of Medical Specialties (abms.org) or state medical board public lookup
- DEA license where applicable (required for controlled substances)
For research peptide vendors (no certification is required, but voluntary accountability signals are meaningful):
- Named third-party testing labs with batch-specific reports published per product
- Independent test results accessible before purchase, not only upon request
- Physical business address and phone number that actually routes to someone
- Cold chain shipping documentation included with the order
- Responsive customer service before the sale, not just after a complaint
No credential makes a provider automatically trustworthy, and the absence of a specific credential doesn't disqualify a newer operation. But the presence of verifiable accountability structures limits the room for things to go wrong. And a business that resists providing verifiable information has chosen that position deliberately.
The July 2026 reclassification: what changes and what doesn't
This context matters to how you evaluate providers right now.
Since late 2023, many popular peptides, including BPC-157, TB-500, CJC-1295, ipamorelin, thymosin alpha-1, and others, have been on the FDA's Category 2 list, a designation that blocks licensed compounding pharmacies from legally preparing them. They've been available only through gray-market research vendors, with all the quality uncertainty that entails.
In April 2026, the FDA removed 12 peptides from Category 2 and scheduled the Pharmacy Compounding Advisory Committee (PCAC) to review BPC-157, TB-500, thymosin alpha-1, and others in July 2026. If those reviews result in inclusion on the 503A bulk drug substances list, licensed compounding pharmacies can legally prepare those compounds under physician prescription.
That's a real shift. It moves compounds out of an unregulated gray market into a system with actual manufacturing standards and medical oversight.
What it doesn't do: generate human clinical trial data for these peptides. The reclassification improves the safety infrastructure. The evidence gaps remain. A compounding pharmacy preparing BPC-157 after reclassification is giving you verified purity and proper manufacturing, not a Phase 3 clinical trial. Understanding what the current legal framework does and doesn't cover before choosing a provider is worth your time.
Before July 2026: any provider claiming pharmacy-grade versions of compounds still on Category 2 is misrepresenting their sourcing. After a positive PCAC review: the right question shifts from "which gray-market vendor has it" to "which licensed pharmacies have added it to their formulary." That's a completely different evaluation process, and the provider checklist above applies to both.
For more on how to interpret the documentation that providers share with you, understanding how to read a peptide COA in detail covers the methodology and what it actually tells you.
Frequently asked questions
It depends on the category. FDA-approved peptides like semaglutide are legal only with a prescription. Research peptides sold with "for laboratory use only" labeling are legal to purchase but not approved for human use. Compounded peptides from a licensed pharmacy are legal with a physician's prescription. Buying injectable peptides from a vendor who ships without any prescription requirement is operating in a gray area that has become an active FDA enforcement target. Several major gray-market suppliers received warning letters in 2024 and 2025.
A research peptide vendor sells compounds labeled "for laboratory use, not for human consumption." No prescription is required and no regulatory manufacturing standards apply. What's in the vial depends entirely on what the vendor chooses to do voluntarily. A telehealth provider, when operating correctly, connects you with a licensed physician who evaluates your health, writes a prescription, and sources from a licensed compounding pharmacy with real quality controls in place. The gap in accountability between those two channels is significant, and not always visible from a website.
A real COA is batch-specific, tied to the lot number you're actually receiving, not a generic document covering all products. It names a testing laboratory you can look up and contact directly. It includes HPLC purity data with the methodology shown, not just a number. And the lab it references should be able to confirm the document is theirs. That last step is the one most buyers skip: contacting the lab directly. If the lab can't find the report in their records, the document is fabricated.
For FDA-approved injectable peptides like semaglutide, yes: they are drugs and prescriptions are required. For compounded peptides from a licensed 503A or 503B pharmacy, yes. For research peptides sold online with "for laboratory use" labeling, technically no, but those compounds are not approved for human use, and the absence of a prescription requirement is not consumer protection. Whether you can obtain them without a prescription and whether you should use them without any medical oversight are two separate questions.
Legally, it means the product is classified as a laboratory reagent, sold for use in test tubes and animal models. It's a seller's liability shield: vendors can sell the compound without the manufacturing standards required for medications, and if something goes wrong, the label is their defense. It says nothing about product quality, purity, or what's actually in the vial. A "research use only" label on something you're planning to inject is not consumer protection: it's the absence of it. The label protects the seller. It does nothing for you.
Published by the PEPVi editorial team. Read our editorial standards.