
The first therapeutic peptide was found in a dog's pancreas, in a Toronto lab, in the summer of 1921. That peptide was insulin. The hundred years between then and now (biohacking subcultures, weight-loss drugs everyone suddenly recognizes, federal reclassification fights) is one of the stranger stories in medicine.
Most people who hear the word "peptide" today assume it's a recent invention. It isn't. Peptides are short chains of amino acids your body already makes by the thousands. What's changed is our ability to synthesize them, our understanding of what they do, and how many people now want access to them outside of traditional medicine.
1921: Insulin and the proof of concept
Frederick Banting was 29, without a lab of his own. He talked his way into lab space at the University of Toronto from a physiology professor named John Macleod, then spent the summer of 1921 extracting something from dog pancreatic tissue alongside a medical student named Charles Best. They called it "isletin" at first. It became insulin, from the Latin for "island," the islets of Langerhans where it's made.
On January 11, 1922, a 14-year-old named Leonard Thompson became the first human to receive it. He'd been in a diabetic coma. Within weeks, he left the hospital. Banting and Macleod won the Nobel in 1923. Commercial production started the same year through Eli Lilly.
Concept to commercial product: 18 months.
What that proved, and this is the part that matters for everything that follows, is that a short-chain amino acid molecule could be isolated from biology, purified, manufactured, and given to a dying person with results that nothing else could achieve. People on insulin didn't just improve. They didn't die.
The quiet decades: building the toolkit (1950s–1980s)
For the next three decades, peptide science moved through academic labs, invisible to most people.
Vincent du Vigneaud synthesized oxytocin in 1953, the first time a peptide hormone had been produced entirely in a lab with no biological source involved. Nobel Prize in Chemistry, 1955. Frederick Sanger completed the first full amino acid sequencing of insulin that same year, Nobel to him too. He'd go on to win a second in 1980, one of only four people who ever have.
The event that actually changed the field came in 1963, when Robert Merrifield published a method called solid-phase peptide synthesis, or SPPS. Before it, building a custom peptide meant months of sequential wet chemistry that could fail at any step. Merrifield's approach assembled amino acid chains on a resin support, link by link, dramatically cutting time and error. Nobel in 1984.
SPPS is still how synthetic peptides are made. The compounds people are currently ordering from gray-market suppliers, debating in Reddit threads, injecting at home: almost all of them were built using a variation of what Merrifield worked out more than 60 years ago.
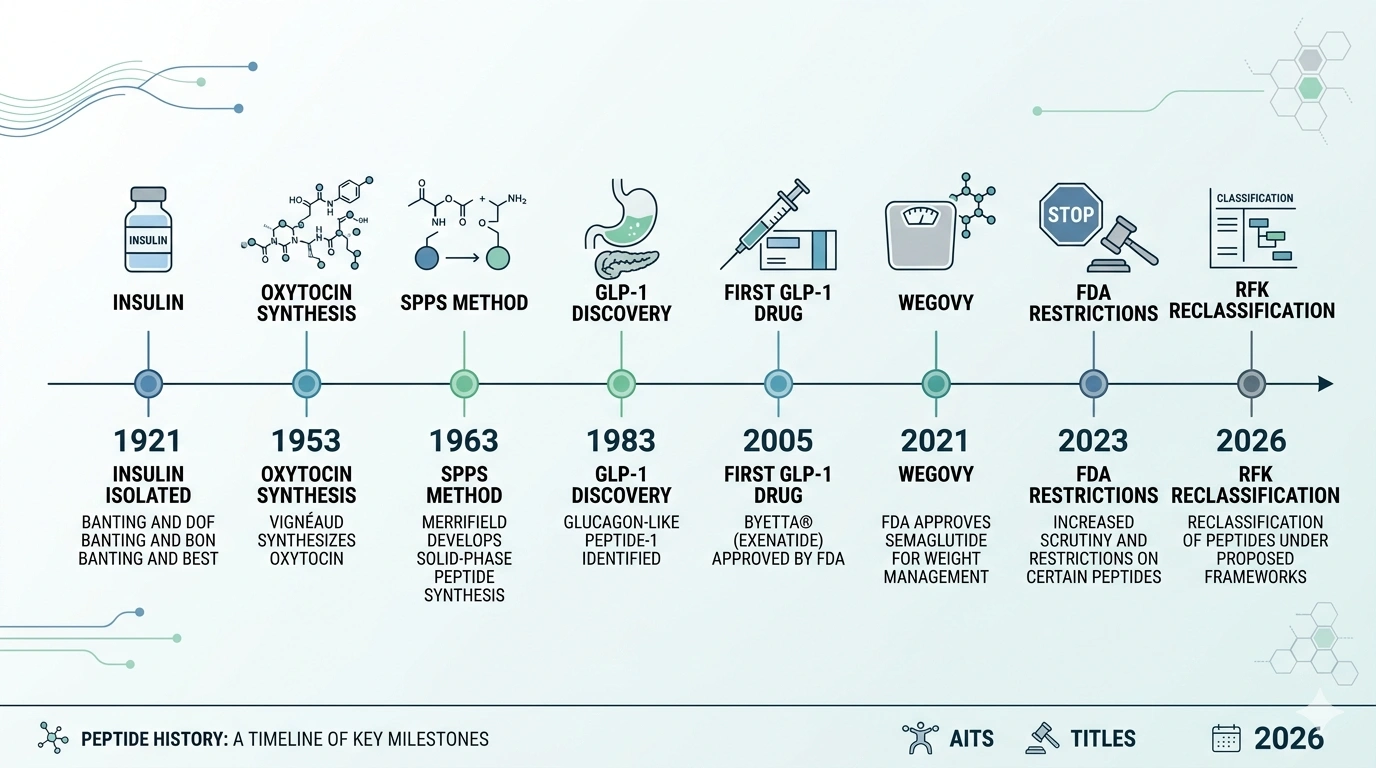
Key milestones in peptide history: from insulin's discovery in 1921 to the 2026 reclassification push.
GLP-1: the peptide that changed the conversation
In 1983, Graeme Bell's team at Chiron Corporation published research in Nature on the proglucagon gene sequence. Buried in it was a fragment they called glucagon-like peptide-1, or GLP-1. Danish physiologist Jens Juul Holst spent much of the next decade working out what it does.
GLP-1 is released from your small intestine after eating. It prompts insulin secretion, signals satiety to your brain, and slows gastric emptying. In type 2 diabetes, that signaling is impaired. Drug researchers started asking if they could engineer a version that lasted longer.
It took about 25 years. Exenatide (Byetta) was the first GLP-1 drug the FDA approved, in 2005. Liraglutide (Victoza) in 2010. Then semaglutide.
Ozempic was approved in December 2017 for type 2 diabetes. Wegovy, the same molecule at a higher dose for weight management, in June 2021. None of this was new science. Holst had mapped the biology 30 years before. What changed was delivery (a formulation stable enough for weekly injection), the trial results, and scale.
Within a few years they were among the most recognized drugs in the country. GLP-1 drugs were a lot of people's first real encounter with a peptide, and most of them just didn't know that's what they were looking at.
The biohacking wave
Before any of that, a smaller community had been running its own experiments.
Bodybuilders were using growth hormone-releasing peptides by the late 1990s. By the early 2010s, a more technically oriented subculture (people tracking their own labs, running cold exposure, stacking supplements) had started experimenting with compounds like BPC-157 and TB-500, mostly sourced from gray-market research chemical suppliers. These needed refrigeration, reconstitution, self-injection. High enough barrier that the practice stayed genuinely niche.
GLP-1 changed the math. When tens of millions of Americans started injecting semaglutide every week without much ceremony, self-injecting a peptide stopped feeling strange. By 2024 some biohackers were using AI tools to research stacks. By early 2025 the trend was showing up in tech and founder circles.
Google searches for "Chinese peptides" went from roughly 35 per month in January 2025 to about 11,000 by January 2026, a roughly 300-fold jump in 12 months. Whether that represents informed consumers or a different kind of problem is a fair question. Regulators noticed either way.
The regulatory collision: 2023–2026
In 2023, the FDA removed 19 peptides from the substances eligible for compounding at licensed 503A pharmacies. Compounding is how a licensed pharmacy makes a custom formulation for a specific patient with a prescription. For most people accessing BPC-157 or TB-500 legally, this was the channel.
The FDA's reasoning: most of these compounds had no randomized controlled trials in humans. Strong animal research in some cases, but not human data. That's not a fabricated concern: animal-to-human translation is genuinely uncertain, and the FDA's job isn't to assume otherwise.
Phase III clinical trials cost tens of millions of dollars. None of these peptides can be patented. There's no company with a financial reason to run those trials, which means some of these compounds may be stuck on the wrong side of the evidence threshold indefinitely, not because they don't work, but because the economics of proving it are impossible.
On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced plans to move approximately 12 of those 19 compounds from Category 2 (restricted from compounding) back to Category 1 (eligible for compounding). The list includes BPC-157, TB-500, Thymosin Alpha-1, CJC-1295, Ipamorelin, AOD-9604, Selank, and Semax. The FDA's Pharmacy Compounding Advisory Committee has a public meeting scheduled for July 23–24, 2026 to formally evaluate whether seven of them should be reclassified.
What reclassification actually means: licensed compounding pharmacies could produce these compounds with a valid physician's prescription. That's meaningful access. It's not FDA approval. It doesn't change the evidence base, and it doesn't produce standardized dosing guidelines. These would still be unapproved drugs — just legally accessible ones through a specific channel.
The July committee meeting is the most consequential formal event in the peptide space in years. Outcomes are genuinely uncertain.
Where things stand
Over 80 peptide drugs have been approved globally as of 2025, with more than 650 in clinical development. The pharmaceutical side of this field is large and growing in a fairly unremarkable way: biotech pipelines, clinical trials, FDA submissions, the usual machinery.
The wellness and biohacking side runs on different logic: faster, less regulated, driven more by community knowledge-sharing and individual experimentation than institutional research. The two aren't completely separate (the GLP-1 story connects them), but they operate on different timelines and different evidence standards.
Insulin is still what it was in 1921. Proof that a peptide could be found, isolated, manufactured at scale, and given to patients with results nothing else could match. The hundred years since have been, in different ways, an attempt to find more molecules that do what insulin did.
The current moment has something the previous 90 years didn't: a large general audience paying attention. That changes what gets asked, what gets funded, and what gets regulated. It doesn't resolve the underlying tensions between access and evidence, between individual experimentation and institutional caution. But it means those tensions now play out in public, and quickly.
Frequently Asked Questions
Insulin, isolated in 1921 by Frederick Banting and Charles Best at the University of Toronto. The first human to receive it was 14-year-old Leonard Thompson, who was near death from type 1 diabetes. He recovered. Commercial insulin production began in 1923 through Eli Lilly, making it the first peptide drug brought to market at scale. Banting and Macleod received the Nobel Prize that same year.
GLP-1 was identified in the early 1980s following the sequencing of the proglucagon gene. Graeme Bell's team published foundational research in Nature in 1983, and physiologist Jens Juul Holst characterized its function over the years that followed. The first GLP-1 receptor agonist drug (exenatide) wasn't FDA-approved until 2005, roughly 25 years after the initial discovery.
In February 2026, HHS Secretary Robert F. Kennedy Jr. announced plans to move approximately 12 peptides from FDA Category 2 (restricted from compounding) back to Category 1 (eligible for compounding). If finalized, licensed 503A compounding pharmacies could produce these compounds with a valid physician's prescription. It's not FDA approval. These peptides would still lack the clinical trial data that full approval requires. The FDA advisory committee meeting in July 2026 is the next formal step.
Bodybuilders were experimenting with growth hormone-releasing peptides in the late 1990s, and a biohacking subculture had been using compounds like BPC-157 and TB-500 since at least the early 2010s. The practice went from genuinely niche to noticeable in the early-to-mid 2020s, partly because GLP-1 drugs normalized self-injection of peptides for a much broader population.
Depends entirely on which peptide you mean. Insulin has been in medical use since 1922. Oxytocin was synthesized in 1953. There are over 80 approved peptide drugs globally. At the same time, many compounds circulating in wellness and biohacking circles (BPC-157, TB-500, Selank) have little or no human clinical trial data and exist in a legal gray zone. "Peptide" spans a huge range, from century-old medicine to experimental compounds with uncertain safety profiles.
Published by the PEPVi editorial team. Read our editorial standards.