BPC-157 is a synthetic peptide with a growing reputation for accelerating healing — tendons, muscles, gut lining, nerves. The animal research is genuinely impressive. The human evidence is genuinely thin.
That gap is the whole story, and it's one most articles gloss over. This guide won't. Whether you've heard about it from a bodybuilder, a biohacker podcast, or your gastroenterologist, you'll find a straight answer here: what BPC-157 actually is, what the research shows, how it's used, and what the legal situation looks like heading into July 2026.
What is BPC-157?
BPC-157 is a synthetic peptide made of 15 amino acids. It was derived from a protein found naturally in human gastric juice: specifically, a portion of that protein that seems to protect the stomach lining from its own acid.
The name comes from that origin: Body Protection Compound, numbered 157 based on the sequence it was isolated from. Researchers at the University of Zagreb, led by Dr. Predrag Sikiric, first characterized it in the early 1990s, and most of the foundational research still comes from that group today.
It's not a natural supplement. BPC-157 doesn't occur in meaningful concentrations anywhere in the body, and you can't get it from food. It's a lab-synthesized compound modeled after a naturally occurring protein sequence. That distinction matters when you're evaluating what it actually is and what claims to believe.
If you're new to peptides entirely, understanding what peptides are and how they differ from proteins and supplements is worth doing before going deeper here.
How BPC-157 works in the body
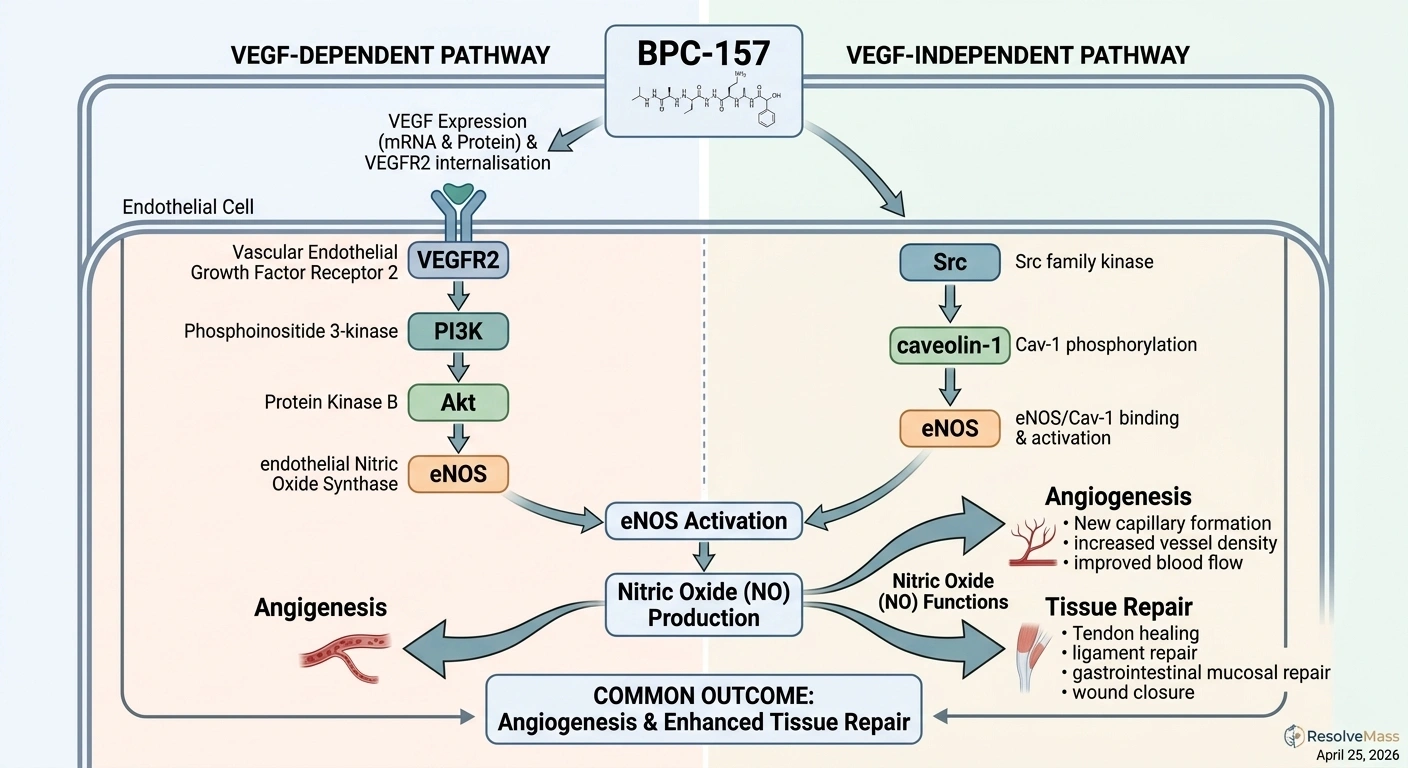
BPC-157 activates several overlapping pathways involved in tissue repair and inflammation control. The most well-characterized involves nitric oxide (NO) signaling and angiogenesis (the formation of new blood vessels).
When BPC-157 is present, it activates VEGFR2 (Vascular Endothelial Growth Factor Receptor-2) through two distinct routes: a VEGF-dependent pathway and a VEGF-independent one running through Src-caveolin-1-eNOS. The result is increased nitric oxide production, which dilates blood vessels, promotes the growth of new capillaries into damaged tissue, and accelerates the delivery of nutrients and growth factors needed for repair.
For tendons specifically, BPC-157 appears to upregulate growth hormone receptors in fibroblasts, the cells responsible for building connective tissue. More GH receptor expression means those cells respond more strongly to the growth hormone already circulating in your body, which speeds the production of new collagen and tendon matrix.
It also reduces oxidative stress by upregulating heat shock proteins and heme oxygenase-1, which protect cell membranes and mitochondria during the inflammatory phase of healing. In the gut, BPC-157 appears to modulate the inflammatory cascade in a way that limits secondary tissue damage.
None of this is established by randomized controlled trials in humans. These mechanisms were identified in animal models and cell culture studies. But mechanistic data is still real data; it tells us how BPC-157 could work, which makes the animal results more plausible, even if they don't confirm human efficacy.
What BPC-157 is used for
Injury recovery: tendons, ligaments, and muscles
This is the primary use case driving most of the interest. BPC-157 has produced consistent results in animal models for tendon tears and ruptures, ligament damage, muscle bruising and tears, and bone healing. That consistency across tissue types and across multiple independent labs is part of what makes the preclinical data worth paying attention to.
Athletes and bodybuilders adopted it for this reason. A stubborn tendon injury that conventional treatment hasn't resolved is a serious problem at competitive levels, and a certain type of person will reach for experimental options if animal data suggests they work. BPC-157 found that population early.
Gut healing
Because BPC-157 was originally isolated from gastric protein, gut applications are the oldest focus in the research. Animal studies show it can heal gastric ulcers, reduce colonic inflammation in IBD models, close GI fistulas (abnormal tissue connections that are notoriously difficult to treat), and support recovery from short bowel syndrome.
The gut healing angle has attracted people with IBD, IBS, leaky gut, and general GI problems. One Phase II clinical trial, run under the name PL 14736 and focused on ulcerative colitis, was completed but never published in peer-reviewed literature. A finished clinical trial that produced no publishable data is a significant gap. It might mean negative results. It might mean regulatory or corporate complications. Nobody outside that research group knows. But it is not a reason for confidence in the gut healing case.
Neurological applications
Later research has examined nerve repair and brain injury protection in animal models. BPC-157 appears to reduce neurological damage from alcohol toxicity, traumatic brain injury, and stroke in rodents. There is also some work on dopamine and GABA pathway interactions.
This area is more speculative than the musculoskeletal and GI applications. The animal data exists, but the translation question is especially open here, and almost no one is pursuing formal trials.
Who's actually using it
The BPC-157 user population falls into a few groups, and the profile matters for evaluating the anecdotal reports that make up most of the available "human" data.
- Athletes and bodybuilders who've hit a tendon or ligament injury that isn't healing. They're often willing to try experimental options and tend to be meticulous about tracking results.
- Biohackers and longevity-focused individuals using BPC-157 as part of broader recovery stacks, sometimes combined with TB-500 or other tissue-repair peptides.
- People with gut conditions (IBD, SIBO, leaky gut) who've exhausted or want to avoid pharmaceutical options and are looking at alternatives.
- Patients with chronic pain or injuries that haven't responded to standard care.
This population skews toward people with higher risk tolerance and experience with self-directed health interventions. Their reports of results are genuine feedback. They're not controlled data; they don't account for placebo effects, other concurrent treatments, or natural healing timelines. But they're not nothing, either, and they've driven serious research interest.
Understanding what peptide therapy actually involves will give you a better framework for evaluating whether BPC-157 fits into a broader treatment plan.
What the research actually shows
Let's be direct: BPC-157 has strong animal data and almost no human data.
A 2024 narrative review in PMC examined 36 studies on BPC-157 for musculoskeletal healing. Thirty-five were preclinical, conducted in rats, mice, or rabbits. One was a human clinical study. Animal-to-human translation is imperfect, and compounds that work dramatically in rodents fail to replicate in humans regularly.
That ratio is not unusual for experimental peptides; animal studies are cheaper and faster to run.
The human evidence that exists is limited but not empty:
- A pilot study on intraarticular injection for chronic knee pain found 7 of 12 patients reported significant pain relief lasting at least six months after a single injection.
- A second pilot study examined BPC-157 for interstitial cystitis.
- A third examined IV administration for safety and pharmacokinetics.
Three small, unblinded pilot studies. None were randomized. None were large enough to draw conclusions from. They're a start, not a foundation.
The unpublished Phase II UC trial is probably the most frustrating data point in the BPC-157 story. The research was conducted. Results were never published. When a completed trial produces no peer-reviewed output, the scientific record has a hole in it that speculation can't fill. It is not evidence for or against efficacy; it's simply missing.
The honest summary: BPC-157 does things in animal models that suggest it could be genuinely useful in humans. Whether it is, in what contexts, and at what doses is unknown.
How BPC-157 is used: oral vs. injectable
This is one of the most practically confusing parts of the BPC-157 conversation, and most sources handle it badly.
Injectable
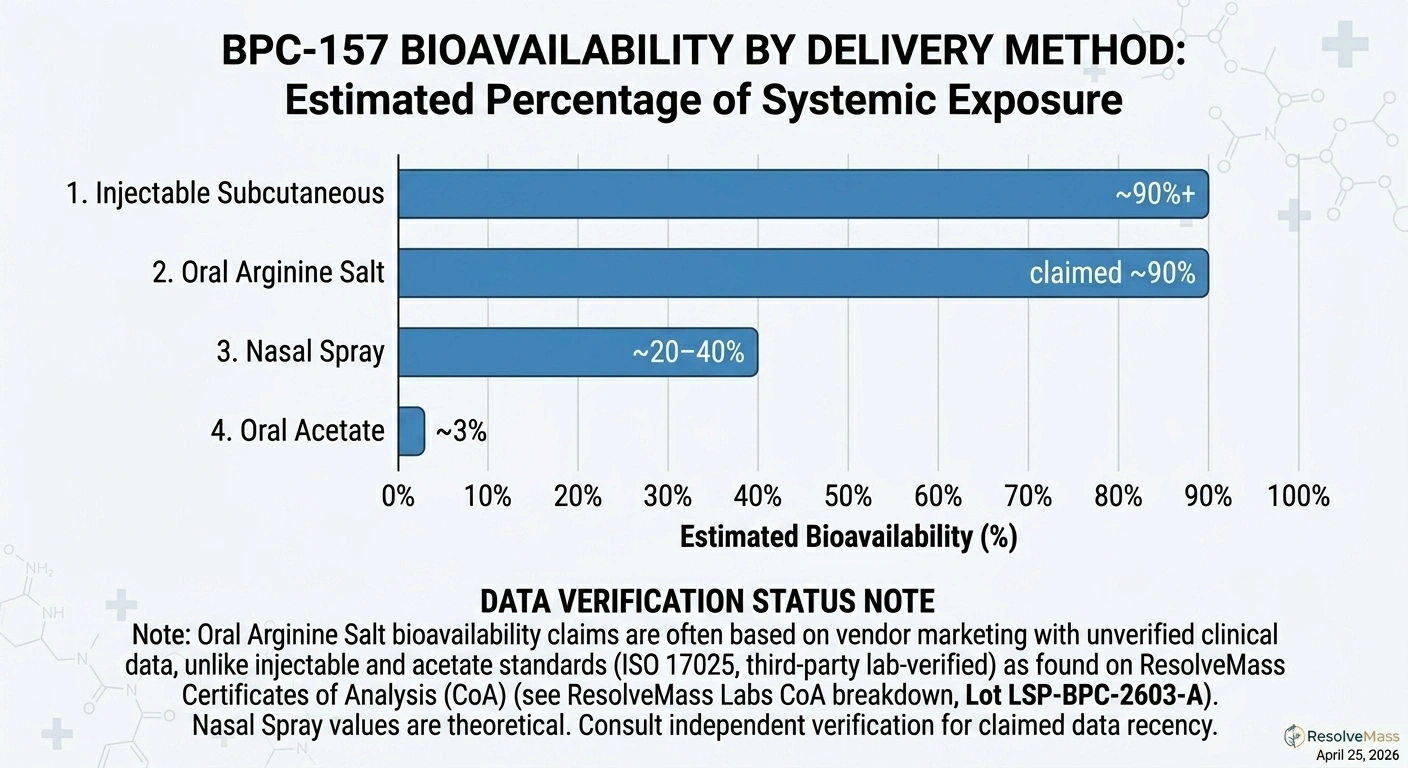
Subcutaneous injection is the most common administration route for musculoskeletal applications. You inject at or near the injury site. Bioavailability via injection is high; the compound reaches the bloodstream quickly and directly.
Intramuscular injection is used in some protocols but less common. Dosing ranges found in community protocols and animal study scaling typically fall between 250 mcg and 500 mcg per dose, once or twice daily. There is no established safe human dose. Those numbers come from animal study extrapolation and anecdotal community protocols, not from human clinical trials. That distinction should inform how much weight you put on any specific dosing recommendation you encounter online.
Oral
Oral BPC-157 is more complicated than most discussions let on.
The standard form (BPC-157 acetate) has very low oral bioavailability in animal models, around 3%. The GI tract degrades it before meaningful systemic absorption can occur. Some researchers argue that for gut-specific applications, oral delivery still makes sense because the compound contacts GI tissue directly regardless of systemic levels. That's a reasonable hypothesis. It hasn't been tested properly in humans.
A stabilized form (BPC-157 arginine salt) claims dramatically better bioavailability (around 90%) in patent data. The stabilization is designed to survive stomach acid. Those claims have not been independently verified in published human research. Patent filings are not peer review.
If you're taking oral BPC-157 for systemic effects and using the standard acetate form, you may be getting very little out of it. If you're taking it for gut applications, it might still be doing something at the tissue level. Nobody has rigorously tested either scenario in humans.
Nasal spray
Nasal administration falls between oral and injectable: estimated bioavailability of 20-40% in animal models. Used less commonly, but the middle-ground profile makes it appealing for people who want to avoid injection.
Side effects and what we don't know about safety
The preclinical safety profile is generally favorable. No significant toxicity has been found at doses used in animal research. Local irritation at the injection site is the most commonly reported issue in animal studies.
Human anecdotal reports from online communities describe nausea, dizziness, injection site reactions, and occasionally anxiety or mood changes. These come from uncontrolled self-reporting, so they can't be interpreted with precision. Rare severe reactions appear in isolated accounts but have no systematic frequency data behind them.
The FDA's formal concern isn't about these short-term effects. It's about immunogenicity: the risk that a synthetic peptide triggers an immune response. Foreign proteins can cause the immune system to produce antibodies against them, which can range from mild reactions to serious autoimmune complications. This hasn't been observed with BPC-157, but it hasn't been adequately evaluated in human studies either.
BPC-157 promotes angiogenesis — the same mechanism tumors use to recruit blood supply and grow. In theory, a pro-angiogenic compound could accelerate tumor growth in someone with undiagnosed cancer. This effect has not been observed in animal tumor models, but the theoretical mechanism is real. For people with cancer history or elevated risk, this is a conversation to have with a physician before considering any pro-angiogenic compound.
What's absent is any long-term human safety data at all, not just long-term, but anything beyond very limited short-term observation. This isn't an argument that BPC-157 is dangerous. It's an argument that the risk profile is genuinely unknown.
Legal status in 2026: where things stand and where they're going
The situation before 2026
In 2023, the FDA placed BPC-157 on its Category 2 list under 503B regulations, prohibiting its use by outsourcing facilities, the large-scale compounding pharmacies that supply physicians across the country. It remained legal to purchase as a research compound through peptide vendors (the DEA doesn't schedule it), but it couldn't be prescribed and filled at a licensed pharmacy.
WADA added it to the prohibited list in 2022. Any competitive athlete in a tested sport cannot use it, regardless of US legal status.
What changed in February 2026
On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced that 14 of the 19 peptides currently under Category 2 restriction (including BPC-157) would be recommended for reclassification to Category 1. The announcement came during a Joe Rogan Experience interview and was subsequently confirmed through official channels.
Category 1 status allows licensed 503A compounding pharmacies to prepare a compound when a physician prescribes it. This is different from FDA approval; BPC-157 would still be an off-label, unapproved drug. But it would move from the regulatory gray market into the formal medical system.
The July 2026 FDA advisory committee meeting
The formal step is a Pharmacy Compounding Advisory Committee meeting scheduled for July 2026. The committee will review whether BPC-157 and other reclassified peptides can be safely produced by compounding pharmacies, with ulcerative colitis listed as an intended use for BPC-157's specific review. The July meeting is the gate that precedes formal reclassification.
What reclassification does and doesn't mean
Coverage of this has been sloppy in several places, so it's worth being specific.
Reclassification to Category 1 means licensed compounding pharmacies can prepare BPC-157 with a physician's prescription. It does not mean FDA approval. It does not mean clinical efficacy has been established. It does not mean it's available over the counter.
The practical difference is real: instead of buying from a research peptide vendor with no regulatory oversight and highly variable quality, a patient could potentially get a prescription from a physician and have it filled at a licensed, inspected pharmacy. Compounding pharmacies are not FDA-manufacturing facilities, but they operate under state pharmacy board oversight and follow USP standards. That's a meaningful quality improvement over the current vendor market.
For information on how to find and evaluate a legitimate provider, see our guide to choosing a peptide provider.
Frequently Asked Questions
It depends on the form and what you're trying to accomplish. Standard oral BPC-157 (acetate form) has very low systemic bioavailability in animal models, around 3%. If you're trying to achieve systemic tissue repair effects, this form is likely much less effective than injection.
For gut-specific issues, oral delivery may still make sense because the compound contacts GI tissue directly regardless of systemic absorption. A stabilized arginine salt form claims much better bioavailability, but those claims haven't been independently verified in published research. If you're using oral BPC-157, the form matters a lot, and the honest answer is that the oral vs. injectable question hasn't been resolved in humans.
As of April 2026, BPC-157 is not FDA-approved for any human use. It's prohibited for compounding pharmacies under Category 2 rules. It is not scheduled by the DEA, which makes purchasing it as a "research compound" from peptide vendors technically legal in the US, but its use in humans is officially unapproved.
That is expected to change after the July 2026 FDA advisory committee meeting. If reclassified to Category 1, licensed pharmacies could prepare it with a physician's prescription. That doesn't make it FDA-approved; it makes it prescribable from a regulated source.
They work through different mechanisms on overlapping tissue types. BPC-157 primarily drives tissue repair through angiogenesis and nitric oxide signaling. TB-500 (Thymosin Beta-4) works through actin polymerization and cell migration. For tendon and muscle healing, the two have complementary effects, which is why they're often used together.
That said, "they have different mechanisms" is not the same as "stacking them is proven to be more effective." The human data on either compound is limited. The human data on using them together is essentially nonexistent. For a full breakdown of how TB-500 works, see our TB-500 guide.
This is a legitimate question. BPC-157 promotes angiogenesis, the same mechanism tumors use to recruit blood supply. Theoretically, a pro-angiogenic compound could accelerate tumor growth in someone with undiagnosed cancer.
This has not been observed in animal tumor models, including studies where cancer was deliberately induced. There is no positive evidence that BPC-157 causes cancer. There's also no adequate human study to rule it out. If you have a history of cancer or known elevated risk, discuss any pro-angiogenic compound with a physician before considering it.
The animal studies typically show measurable differences within one to four weeks at the tissue level. Community reports from human users suggest some people notice changes in injury recovery within two to four weeks, with gut-related effects sometimes appearing sooner.
There is no clinical timeline data from human trials. The useful variables (injury type, severity, dosing form and quality, individual physiology) all affect how quickly anything changes. Anyone providing a confident specific timeline is extrapolating from animal data or anecdote.
Published by the PEPVi editorial team. Read our editorial standards.