
TB-500 is a synthetic peptide fragment of thymosin beta-4, a protein your body already makes. Most people find it through recovery contexts (tendons, muscle strains, persistent joint inflammation) and it almost always shows up in the same conversation as BPC-157. Here's what it actually is, how the mechanism works, and where the evidence honestly stands.
TB-500 and thymosin beta-4 are not the same molecule
Most articles use the names interchangeably. They're not the same thing.
Thymosin beta-4 (TB4) is a naturally occurring 43-amino-acid peptide present in virtually every cell in your body. It concentrates in platelets, white blood cells, and wound fluid, and the body releases it as part of the initial repair response after injury. TB4 has multiple functional regions, each with distinct roles in cellular biology.
TB-500 is a synthetic version of just seven of those 43 amino acids: the sequence Ac-LKKTETQ, positions 17 through 23 of TB4. That's the actin-binding domain, the region researchers believe drives most of the tissue repair activity. The full 43-amino-acid protein is harder to synthesize and less stable, so isolating this fragment was a practical choice. Whether the fragment fully replicates the parent molecule's effects is still being worked out.
Most published research on thymosin beta-4 used the full-length protein, not the fragment. When you see a study cited for TB-500's wound healing or anti-inflammatory effects, check which form was actually tested. They're assumed to work similarly; that assumption hasn't been confirmed in head-to-head human trials.
A 2024 study in the Journal of Chromatography B added another layer: the compound that increased wound-healing activity in vitro wasn't TB-500 itself but a metabolite, Ac-LKKTE, formed when TB-500 breaks down in tissue. This doesn't mean the effects aren't real; it means the active species may be one step downstream. Most beginner content on TB-500 skips this entirely.
Before getting into the mechanism, the beginner's guide to what peptides are covers the fundamentals if you're new to the subject.
How TB-500 works
The mechanism is actin regulation. That's actual molecular biology, not a marketing reframe, and it explains why TB-500 shows up across multiple tissue types rather than acting on a single target.
Actin sequestration
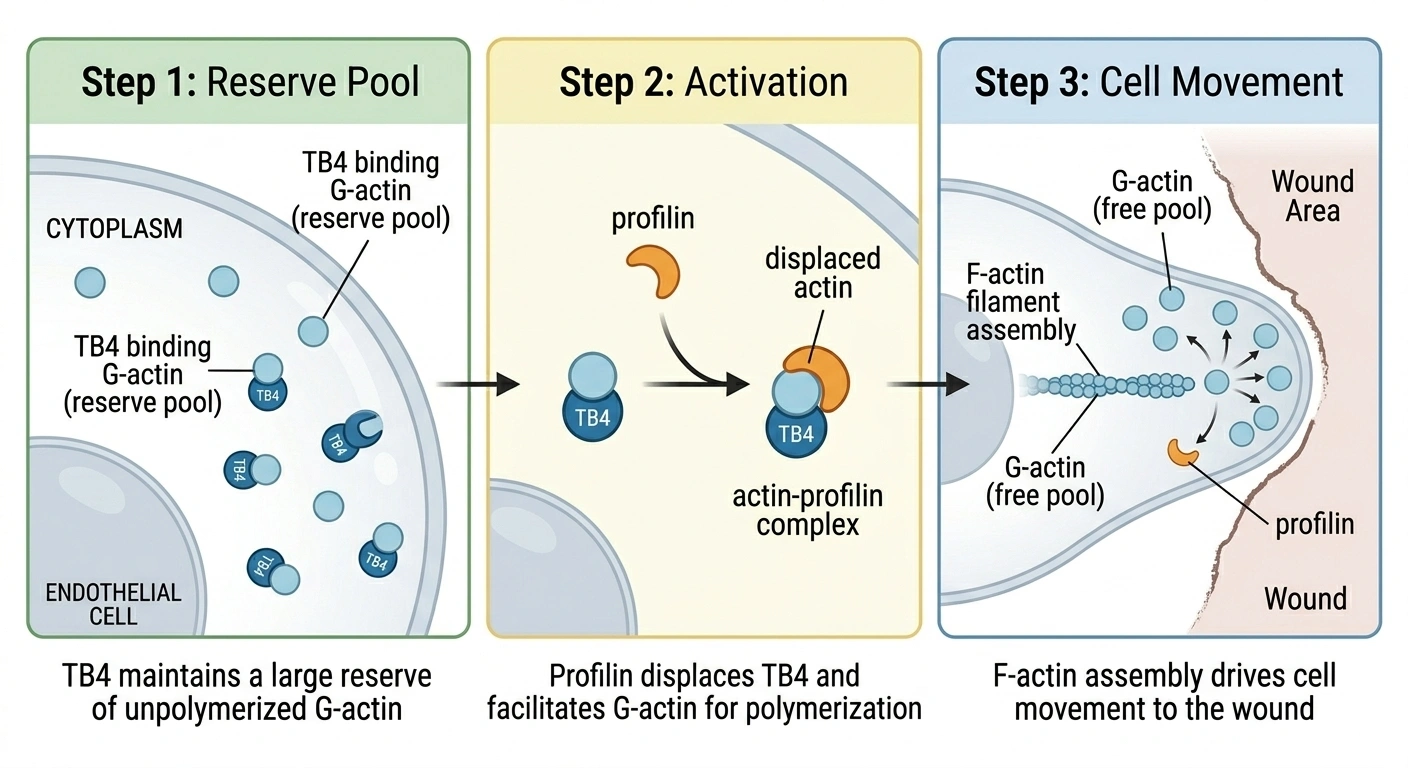
Your cells contain actin in two forms: G-actin (individual monomers, soluble) and F-actin (long filaments that form the structural scaffolding inside cells). The balance between these two forms shifts constantly based on what the cell needs to do.
Thymosin beta-4 is the principal actin-sequestering protein in most mammalian cells. It binds G-actin one-to-one, holding individual monomers in reserve. The protein contacts both faces of each monomer simultaneously, blocking it from joining a growing filament. Think of it as a molecular parking lot: G-actin held ready but inactive, available the moment a cell needs to mobilize.
When a cell needs to move, that reserve matters. Actin polymerization (assembling G-actin monomers into F-actin filaments) is what drives cell movement. A fibroblast crawling toward a wound, an immune cell arriving at an injury site: both depend on rapid filament assembly at the cell's leading edge. TB4's buffer makes that process more responsive by keeping monomers pre-loaded.
The switch from "stored" to "building" happens through profilin, a competing actin-binding protein. Profilin can displace G-actin from its TB4 complex and catalyze the nucleotide exchange that makes monomers polymerization-ready. The TB4/profilin ratio is the control point for when and where actin assembly happens in a cell. TB-500, as the actin-binding fragment, operates through this same relay.
Cell migration and tissue repair
Healing at the cellular level is fundamentally a movement problem. Repair cells have to reach the injury. Fibroblasts have to arrive and build connective tissue. Epithelial cells have to migrate to close a wound. All of it depends on cells reorganizing their cytoskeletons and moving, which is what actin dynamics enable.
Beyond the actin system, TB4 also binds integrin-linked kinase (ILK) at the leading edge of migrating cells, activating a pathway through Akt2 that increases metalloproteinase production. Metalloproteinases digest the extracellular matrix, the protein mesh surrounding cells, clearing a physical route for repair cells to move through damaged tissue.
The animal data gives some sense of the effect size. In diabetic mouse wound models (which heal poorly by design), TB4 treatment accelerated reepithelialization by 42% at day four and up to 61% by day seven compared to untreated controls. That's a large effect in a model chosen specifically because it resists healing.
Angiogenesis and inflammation
TB4 also promotes new blood vessel formation by upregulating VEGF through Notch signaling and activating endothelial progenitor cells. New vasculature is the delivery system for healing: oxygen and immune cells in, metabolic waste out. Tendons have notoriously poor blood supply, which is most of why tendon injuries are so slow to resolve. TB-500's angiogenic activity directly targets that bottleneck.
On the inflammatory side, TB4 blocks NF-kappaB activation, which suppresses downstream cytokines including TNF-alpha and IL-1 beta. There's also evidence for a secondary mechanism: the oxidized form of TB4, produced naturally at sites of inflammation, disperses neutrophils and blocks their chemotaxis. The reduced form sequesters actin; the oxidized form manages the inflammatory response. Consumer articles typically compress this to "anti-inflammatory," but the biology involves at least two distinct programs running in parallel.
What TB-500 is used for
Because the mechanism touches cell migration, angiogenesis, and inflammatory signaling simultaneously, TB-500 gets applied to a lot of conditions rather than one specific target.
- Tendon and ligament injuries are the most common reason people research TB-500. Slow tendon healing comes down to poor vascularity. The angiogenic effect addresses that directly by promoting capillary growth into healing tendon tissue. For a broader look at how peptide options compare for tendon recovery, the tendon healing guide covers the evidence across different compounds.
- Muscle strains are another primary use case. After skeletal muscle injury, local TB4 production increases naturally and the peptide acts as a chemoattractant for myoblasts, the progenitor cells responsible for muscle regeneration. A 2010 study found that both TB4 and its sulfoxide form significantly accelerated myoblast migration and wound closure in experimental models.
- Chronic wounds and post-surgical healing have some of the strongest preclinical evidence, particularly in impaired healing models. The Phase 2 clinical work that has been completed is largely in wound indications, not sports medicine, which is worth noting when you see wound healing studies cited to support athletic recovery use.
- Joint pain and chronic inflammation come up frequently, particularly for injuries that haven't fully resolved. The mechanism supports this application. Controlled human data for this specific use is sparse.
- Hair growth is legitimately studied, not gym-forum lore. Multiple animal studies from 2003 through 2019, from independent research groups, documented TB4 promoting hair follicle development via stem cell mobilization and improved vascular supply around follicles. No human trial has run yet. The animal evidence is consistent enough to take seriously, but "consistent animal data" and "proven to regrow your hair" are different claims.
- Cardiac repair is where the research gets genuinely interesting, well outside the typical recovery context. Studies show TB4 can reduce myocardial cell death after a heart attack, promote vascular regrowth in cardiac tissue, and activate dormant cardiac progenitor cells. Phase 2 human work has been conducted. Most people reading this aren't using TB-500 for cardiac purposes, but it explains why the peptide gets serious scientific attention rather than just biohacker attention.
For more context on where TB-500 fits within recovery peptide options, the muscle recovery guide compares it alongside related compounds.
TB-500 vs. BPC-157
These two get paired constantly. Both are research peptides, both show up in recovery contexts, both are WADA-banned. They work through different biology and aren't doing the same job.
BPC-157 is 15 amino acids derived from a gastric juice protein. TB-500 is 7 amino acids from thymosin beta-4. Their structures share nothing.
BPC-157 works primarily through VEGFR2, a growth factor receptor, and directly stimulates nitric oxide production via eNOS. It's particularly effective at promoting collagen organization in tendons and significantly upregulates growth hormone receptors at injury sites. The effects are more localized; BPC-157's strongest data is for gut healing, tendon repair, and wound healing at or near the application site.
TB-500 is more systemic. Inject it anywhere and it supports cell migration and angiogenesis throughout the body, not just at the injection site. TB-500 has stronger data for cardiac repair and anti-fibrotic effects (reducing scar tissue formation in liver and kidney models). BPC-157 has essentially no comparable cardiac evidence. For gut problems and tendon healing specifically, BPC-157 has more direct preclinical support.
These aren't competing. Different mechanisms, different strengths, genuinely complementary. The rationale for stacking them is that BPC-157 handles localized tissue-level repair and growth factor signaling while TB-500 supports the systemic cell migration and angiogenesis that delivers repair resources to the site. The combination gets called the "Wolverine stack." No controlled clinical trial has studied it; the evidence for stacking comes from mechanistic inference and community experience. Full breakdown in the BPC-157 guide.
What the research actually shows
Don't trust any source that presents the TB-500 evidence as settled. It isn't.
The preclinical case is strong. Multiple independent labs have replicated wound healing, angiogenesis, and cell migration findings across animal models and tissue types. The mechanism is structurally characterized: crystal structures of the TB4/actin complex exist. That's a higher bar than most research peptides meet.
Human safety data is real. Two Phase 1 trials tested intravenous thymosin beta-4 in healthy volunteers at escalating doses: one in the US (2010), one in China (2021). Both reported no serious adverse events. Phase 2 trials for wound healing and eye disease have been conducted. That safety record is genuinely meaningful.
What doesn't exist: randomized controlled trials for the musculoskeletal injuries most people actually use TB-500 for. Tendons, rotator cuffs, muscle tears, sports injuries: no RCT has been published for any of them. The Phase 2 work is for wound healing and corneal repair, not athletic recovery. The extrapolation from one to the other is mechanistically reasonable; it hasn't been tested in humans.
The 2024 metabolite finding is an active research question rather than a verdict, but it adds honest complexity: if the active species in tissue is Ac-LKKTE and not TB-500 itself, that's relevant to how researchers should think about dosing and design future compounds.
A 2026 SAGE Journals study examined Reddit users who had used peptides after orthopedic surgery. Self-reported outcomes were largely positive. Self-reported data without controls proves nothing about efficacy, but it documents that large numbers of people are using TB-500 outside clinical settings and reporting subjective improvement. Context, not evidence.
Honest summary: well-documented mechanism, strong animal evidence, real Phase 1 safety data, no efficacy RCTs for the specific injuries driving most consumer interest. Anyone claiming certainty in either direction is simplifying.
Legal status in 2026
TB-500's regulatory situation shifted in early 2026, but not as cleanly as some coverage suggests.
In September 2023, the FDA classified thymosin beta-4 fragment as a Category 2 bulk drug substance, which blocked licensed US compounding pharmacies from preparing it. That ended the physician-supervised access pathway. Demand shifted to unregulated research chemical vendors, precisely the outcome the FDA regulation was supposed to prevent.
On February 27, 2026, HHS Secretary RFK Jr. announced that approximately 14 of the restricted peptides, including TB-500, would be moved back to Category 1. The announcement acknowledged directly that the Category 2 restrictions had driven demand toward less safe sources. On April 15, 2026, HHS formally directed removal of 12 peptides from Category 2.
The important caveat: this doesn't restore legal access yet. The Pharmacy Compounding Advisory Committee (PCAC) is scheduled to review TB-500's case on July 23-24, 2026. Until that process concludes and the FDA formally updates its list, compounding pharmacies cannot prepare TB-500 under Section 503A. The public comment docket (FDA-2025-N-6895) remains open through July 22, 2026. Legal compounding access is expected — it's not here yet.
Anyone currently using TB-500 is sourcing it from research chemical vendors. No regulatory oversight on purity, dosing accuracy, or contamination. That's a real gap.
For competitive athletes: WADA lists TB-500 as a non-specified substance — meaning a standard four-year ban for a first offense, not the reduced two-year ban that applies to specified substances. It is banned in- and out-of-competition. US compounding regulations have no bearing on WADA status.
For a full picture of what physician-supervised peptide access looks like through a compounding pharmacy, the guide to peptide therapy covers the clinical pathway in depth.
Frequently Asked Questions
No, but they're closely related. Thymosin beta-4 is the full 43-amino-acid protein your body produces naturally. TB-500 is a synthetic version of just seven of those amino acids, specifically the actin-binding domain (Ac-LKKTETQ, positions 17-23). The two are assumed to work similarly because TB-500 contains the region thought to drive the repair effects, but most published research used full-length TB4, not the fragment. When you see clinical study data cited for TB-500, check which form was actually tested.
Yes, and many people do. They work through different mechanisms: TB-500 through systemic actin regulation and cell migration, BPC-157 through localized growth factor signaling and collagen organization. So they address different aspects of healing rather than duplicating each other. The combination is called the Wolverine stack. No known interaction risk exists between them. There are also no controlled clinical trials of the combination; everything about stacking comes from mechanistic inference and community experience, not controlled data.
There's no FDA-approved dosing protocol because there's no approved indication. What circulates in clinical and research settings typically looks like 2 to 2.5 mg twice weekly during a loading phase of four to six weeks, followed by 2 mg once per week or every two weeks for maintenance. Cycles usually run six to twelve weeks. These figures come from practitioner experience and community protocols. The Phase 1 safety studies used intravenous administration, which has different pharmacokinetics than subcutaneous injection, so those doses aren't directly comparable.
Evidence on timing comes from wound healing studies and self-reported experience rather than controlled injury trials. Animal and clinical wound studies suggest reduced inflammation and early healing improvements can appear within one to two weeks. Structural repair (tendon healing, muscle tissue rebuilding) typically takes four weeks or more, with full courses running eight to twelve weeks. No randomized controlled trial has specifically measured this timeline for the sports injuries most people use TB-500 for.
It's in transition. TB-500 is not FDA-approved for any indication. The Category 2 restriction blocking US compounding pharmacies from preparing it is being removed following the HHS announcement in February 2026 and formal direction in April 2026, but legal compounding access requires the PCAC review scheduled for July 23-24, 2026 to conclude first. Until then, the clinical pathway isn't open. TB-500 is currently available from research chemical vendors, which operate without regulatory oversight on purity or dosing accuracy. Athletes competing under WADA jurisdiction: the peptide remains banned regardless of what US compounding rules say.
Published by the PEPVi editorial team. Read our editorial standards.