
Peptide therapy is the clinical use of prescription peptides under physician supervision: lab work upfront, a pharmaceutical-grade compound from a licensed compounding pharmacy, and ongoing monitoring as part of the deal. That's what separates it from buying peptides online and dosing yourself, which sits in a very different category of risk and accountability.
Both paths exist. This guide covers the clinical side: what peptide therapy actually looks like in practice, what it's used for, what it costs, and how to find a qualified provider.
Peptide therapy vs. self-administering peptides
The phrase "peptide therapy" gets used loosely online, but in a medical context it has a specific shape. A licensed clinician evaluates your health history, orders labs, and writes a prescription for a peptide compounded at a licensed pharmacy. The compound ships to your home or is administered in-clinic. The prescribing clinician monitors your response and adjusts your protocol over time.
Self-administered peptides, sourced from research chemical vendors, overseas suppliers, or gray-market websites, skip that framework entirely. No physician evaluation, no compounding standards, no quality verification. The price is often dramatically lower. So is the accountability for what's actually in the vial.
Many people self-administer peptides. That's a real thing. But it's a meaningfully different risk profile from working through a clinical program, and it's worth naming that distinction before going further.
If you're starting from scratch on the basics, the beginner's guide to what peptides are covers what peptides are chemically and how they differ from proteins. The explainer on how peptides signal your cells covers the receptor-binding mechanics that make clinical applications possible in the first place.
What conditions is peptide therapy used for?
Peptide therapy doesn't map onto one condition or goal. Clinicians prescribe different peptides for different purposes, and the evidence quality behind each varies considerably.
Growth hormone support is one of the most common entry points. Peptides like Ipamorelin and CJC-1295 stimulate the pituitary to release more of its own GH rather than introducing synthetic growth hormone directly. Most people prescribed these are dealing with age-related decline, looking for improvements in sleep quality and body composition. They've been a staple of anti-aging and functional medicine programs for years.
Injury recovery is the other big category. BPC-157 has strong preclinical data for tendon, ligament, and gut healing. Human clinical trials are still limited, but sports medicine physicians prescribe it widely. TB-500 works through similar mechanisms and shows up in similar contexts.
On the immune side, Thymosin Alpha-1 holds FDA-approved status in several countries for hepatitis B treatment and gets prescribed off-label in the US for immune support in post-infection recovery and some autoimmune contexts.
For metabolic health, GLP-1 receptor agonists like Semaglutide are, technically, peptides, FDA-approved with solid clinical trial data. Beyond GLP-1s, Tesamorelin (FDA-approved for HIV-associated lipodystrophy) and AOD-9604 (human evidence is thin) appear in fat metabolism protocols.
And then there's skin and collagen. Peptides like GHK-Cu are prescribed in some clinical settings for collagen support and wound healing, both topically and systemically.
The evidence across these categories ranges from well-established (GLP-1 peptides) to preliminary-but-promising (BPC-157) to mostly theoretical. A credible provider will tell you which bucket applies to what they're prescribing. If the pitch sounds uniformly confident across every application, that's a signal worth paying attention to. For a goal-by-goal breakdown of what studies actually show, the evidence review on peptide benefits goes deeper.
What a peptide therapy consultation looks like
Whether you're seeing someone in person or via telehealth, a well-run program follows a similar sequence.
You start with an intake: a health history covering medications, diagnoses, lifestyle, and what you're trying to accomplish. This step matters because some peptides are contraindicated in certain conditions. Active cancer is the most commonly cited: several peptides that stimulate tissue growth should not be used by people with certain cancer histories without careful medical review.
Before writing any prescription, a competent provider orders bloodwork. Exactly which labs depends on your goals: hormone panels, metabolic markers, inflammatory markers, or imaging if injury recovery is the focus. Lab costs typically run $200-$500 out of pocket.
The initial consultation itself runs 30-60 minutes, video or in-person. The clinician reviews your labs and history, discusses your goals, and proposes a protocol: which peptide, what dose, what delivery method, and for how long. Most clinical protocols use subcutaneous injection, a small needle into the fat just under the skin, similar to insulin administration.
The prescription goes to a licensed compounding pharmacy. For telehealth programs, the peptide ships to your address with injection supplies and instructions. In-clinic programs may administer initial doses on-site.
Follow-up is where lower-quality programs cut corners. A properly run protocol includes follow-up labs and check-ins at 6-8 weeks to assess response and adjust. Budget for this ongoing oversight; it's part of what you're paying for with a clinical program, and it's what separates that program from just buying a compound online and guessing.
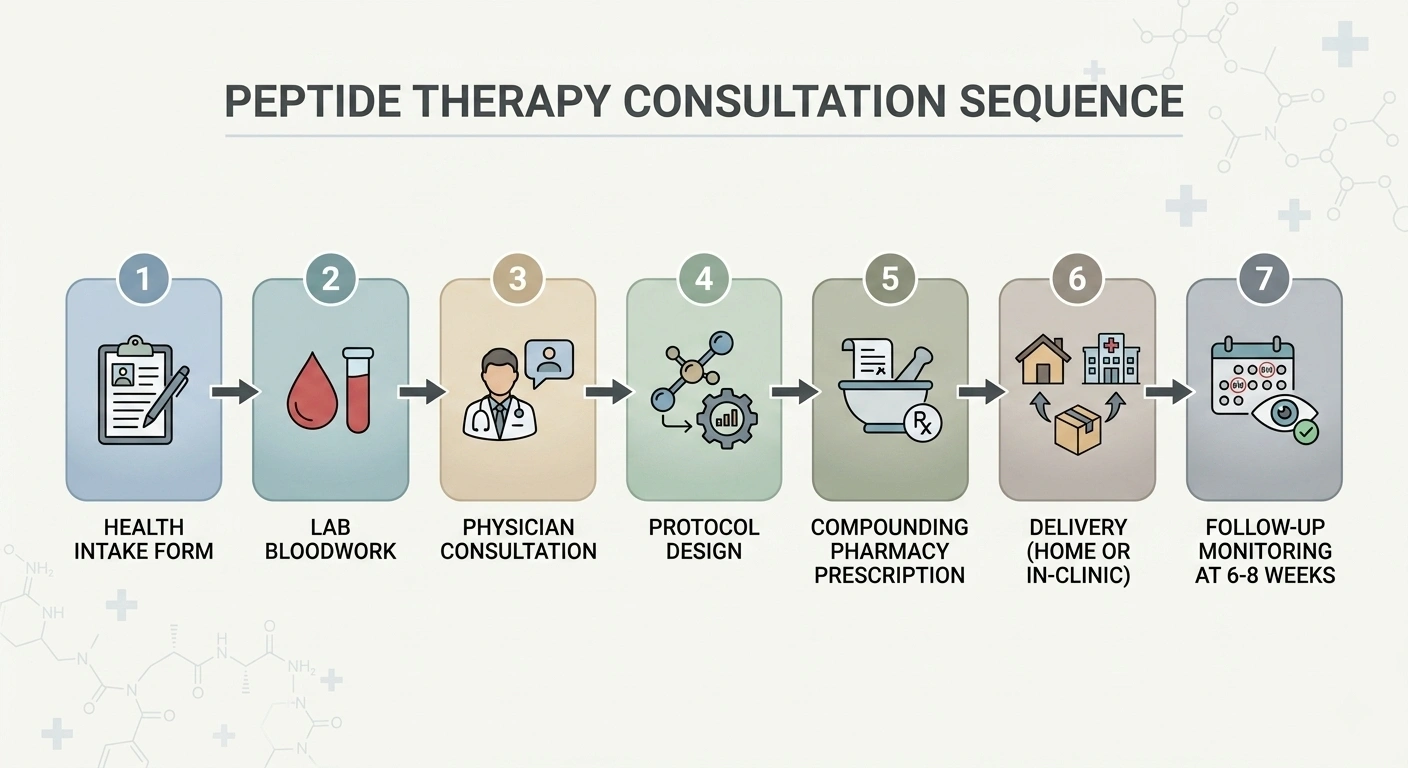
Peptide therapy consultation flow: intake, labs, physician consult, protocol, dispensing, and follow-up.
How much does peptide therapy cost?
Insurance doesn't cover peptide therapy. It's classified as experimental and elective, so everything is out of pocket.
Monthly peptide costs vary by compound and protocol:
- Growth hormone peptides (Ipamorelin, CJC-1295): $200-$450/month
- Healing peptides (BPC-157, TB-500): $100-$300/month
- Immune peptides (Thymosin Alpha-1): $200-$400/month
- Comprehensive hormone protocols: $500-$2,000/month
Those numbers are for the peptide itself. A realistic view of first-year costs adds the initial consultation ($100-$300), lab work ($200-$500 per panel), follow-up visits ($50-$150 each), and shipping ($15-$75 per order). A single, well-managed peptide protocol realistically lands in the $2,000-$5,000 range in year one.
Gray-market vendors often sell the same compounds for $10-$30 per vial. Those are research chemicals not manufactured for human use, with no compounding standards and no accountability for contents. The price gap is real and reflects the difference in oversight, not just a markup.
FDA testing found incorrect dosages or undeclared ingredients in up to 40% of online peptide samples.
Finding peptide therapy: near you and online
"Peptide therapy near me" makes sense as a starting point. In practice, geography matters less than it used to. Telehealth has substantially widened access.
For local in-person providers, the most reliable resource is the International Peptide Society's practitioner directory, searchable by specialty and location. Functional medicine practices, integrative health clinics, and sports medicine physicians are the most consistent sources. Medical spas and concierge clinics also offer peptide therapy, though the clinical depth varies considerably.
If local options are limited, several telehealth platforms now offer licensed physician consultations with home delivery of compounded peptides. Nava Health, HydraMed, and Aspire Health are among the established options. Initial video consultation, prescription, peptide shipped directly to you, follow-up by video or phone.
Two verification steps apply regardless of how you find a provider: confirm the prescribing clinician holds an active license in your state, and confirm prescriptions go to a compounding pharmacy that follows USP 797 sterile compounding standards. These aren't bureaucratic boxes. They're what ensure what you receive matches what was prescribed.
Telehealth vs. in-person clinics
For most peptide protocols, telehealth is a legitimate option. The prescribing decision is based on labs and health history, not a physical exam. Those are as reviewable over video as in person, and ongoing management works the same way.
In-person has a real edge in specific situations: when you want hands-on injection technique training for your first doses, when your evaluation warrants a physical exam, or when the complexity of your situation calls for a more thorough workup.
Telehealth's main practical limit is state licensing: a provider can only prescribe where they hold an active license. Check coverage before you go through an intake process.
Some practices use a hybrid approach: in-person for the initial evaluation, telehealth for ongoing management. That captures most of the convenience without sacrificing the thoroughness of a proper first visit.
What's changing in 2026
The regulatory situation around peptide therapy has been in flux, and 2026 is a significant year for it.
In 2023, the FDA placed 19 commonly used peptides on a Category 2 restricted list, effectively ending their legal use through US compounding pharmacies. BPC-157, Ipamorelin, TB-500, KPV, GHK-Cu, and others disappeared from clinical programs overnight. Many patients either stopped treatment entirely or turned to gray-market sources.
On February 27, 2026, HHS Secretary RFK Jr. announced that approximately 14 of those 19 peptides would be moved back to Category 1 status. As of April 2026, the FDA confirmed it will remove 12 peptides from the restricted list, though the formal updated list had not yet been published at time of writing. Compounds expected to return to legal compounding access include BPC-157, Thymosin Alpha-1, TB-500, Ipamorelin, Selank, Semax, KPV, MOTS-C, GHK-Cu, and others.
Two things worth being clear on: moving to Category 1 is not FDA approval. These remain off-label therapeutics. The reclassification restores the legal pathway for licensed compounding pharmacies to prepare them with a physician's prescription. Clinical oversight remains required.
Five peptides are expected to stay restricted due to more serious safety concerns: Melanotan II (skin cancer and cardiovascular risks), GHRP-2, GHRP-6, CJC-1295 (cardiac side effect data), and LL-37 (insufficient human safety data).
If you've been waiting to start a clinical protocol, or if your provider lost access to specific compounds in 2023, the second half of 2026 should meaningfully expand your options through legitimate clinical channels.
Questions to ask before you start
Working with a provider means evaluating that provider. The basic questions on the clinical side: are they a licensed physician, nurse practitioner, or physician assistant? Are they licensed in your state? How much of their practice involves peptide therapy specifically? And do they order labs before prescribing, or do they offer protocols to patients without reviewing bloodwork first?
On the peptide itself, ask what the clinical evidence looks like for your specific goal, not just "does this peptide work," but what the evidence actually shows for your intended application. Ask which compounding pharmacy they use and whether it's USP 797 compliant. Ask what side effects to watch for and what to do if they occur.
On the program: what does follow-up look like, and when will you review labs? What are the criteria for stopping or adjusting? If a peptide you're interested in is still under Category 2 restrictions, how is it being sourced?
A provider who answers these directly and without irritation is probably running a real clinical program. One who brushes past them or treats the questions as an obstacle — that's information.
Frequently asked questions
Under physician supervision with pharmaceutically compounded peptides from a licensed pharmacy, the documented risks are relatively limited: mostly injection site reactions, transient headaches, and some water retention with hormone-stimulating peptides. The bigger unknowns are long-term effects, since most non-FDA-approved peptides haven't been through multi-year human safety trials.
The risk profile changes substantially with sourcing. Gray-market peptides from unregulated vendors introduce contamination, incorrect dosing, and undeclared ingredients as real possibilities. FDA testing found incorrect dosages or undeclared ingredients in up to 40% of online peptide samples. A licensed provider paired with an accredited compounding pharmacy eliminates most of that variable. For a safety breakdown by peptide type, the safety guide covers it further.
For a single peptide protocol, expect $150-$450/month for the peptide itself. That doesn't include the initial consultation ($100-$300), lab work ($200-$500 per panel), follow-up visits ($50-$150 each), or shipping. Realistically, a properly managed protocol costs $2,000-$5,000 in the first year. Comprehensive anti-aging or hormone optimization programs run higher. None of this is covered by insurance; peptide therapy is classified as experimental and elective.
The most reliable starting point is the International Peptide Society practitioner directory, which lists board-certified practitioners searchable by location and specialty. Functional medicine practices and integrative health clinics are consistent local sources. If local options are limited, telehealth providers like Nava Health and HydraMed offer licensed physician consultations with compounded peptides shipped to your address, covering most US states.
In February 2026, HHS announced approximately 14 of the 19 peptides restricted by the FDA in 2023 would return to legal compounding status. As of April 2026, the FDA confirmed 12 will be removed from the Category 2 restricted list; the formal updated list was still pending publication at time of writing. Compounds expected to return include BPC-157, Thymosin Alpha-1, TB-500, Ipamorelin, Selank, Semax, KPV, MOTS-C, and GHK-Cu. These will be available through licensed compounding pharmacies with a physician's prescription, not over-the-counter.
For clinical peptide therapy through a licensed compounding pharmacy, yes. Research chemical vendors sell peptides without prescriptions, but those products aren't manufactured for human use and carry no quality guarantees. The prescription requirement isn't just regulatory paperwork; it's what ensures a clinician reviews your situation, checks for contraindications, orders appropriate labs, and monitors your response over time. After the 2026 reclassification, more peptides will move back into the prescription-through-compounding pathway. For all its friction, that structure is substantially safer than the alternative.
Published by the PEPVi editorial team. Read our editorial standards.