
Peptides work by sending signals to your cells. A peptide travels to a target cell, binds to a receptor, and that binding triggers a reaction inside. This page explains what those messages say, how cells receive them, and why the delivery route determines whether any of it actually lands.
Your cells are already running on peptide signals
Your body makes thousands of peptides right now. They regulate hunger, trigger healing, release hormones, dampen inflammation, and keep tissues communicating. All of them share the same basic mechanism, and that's why therapeutic peptides can reach into so many of those same systems.
Think of a peptide as a text message, and the receptor as the phone that receives it. The peptide doesn't force anything. It sends a signal, and the cell responds based on its own programming. If the right receptor picks it up, the cell acts. No matching receptor, nothing happens.
Compare that to anabolic steroids, which flood the body and directly override hormone levels. Peptides don't do that. They work inside the existing system, amplifying signals, triggering processes the body already knows how to run, filling in for something that's dropped off with age or injury.
Not sure what peptides actually are yet? Start with the beginner's guide before getting into mechanics.
How peptides find the right receptor
Not all receptors accept all peptides. The interaction is specific.
Specificity comes from the amino acid sequence. Each amino acid contributes a particular shape and charge. Together they form a three-dimensional structure that either matches a given receptor's binding pocket or doesn't. When the fit is right, the peptide docks, and binding triggers a conformational change: the receptor physically shifts its shape, which activates proteins on the inner membrane surface.
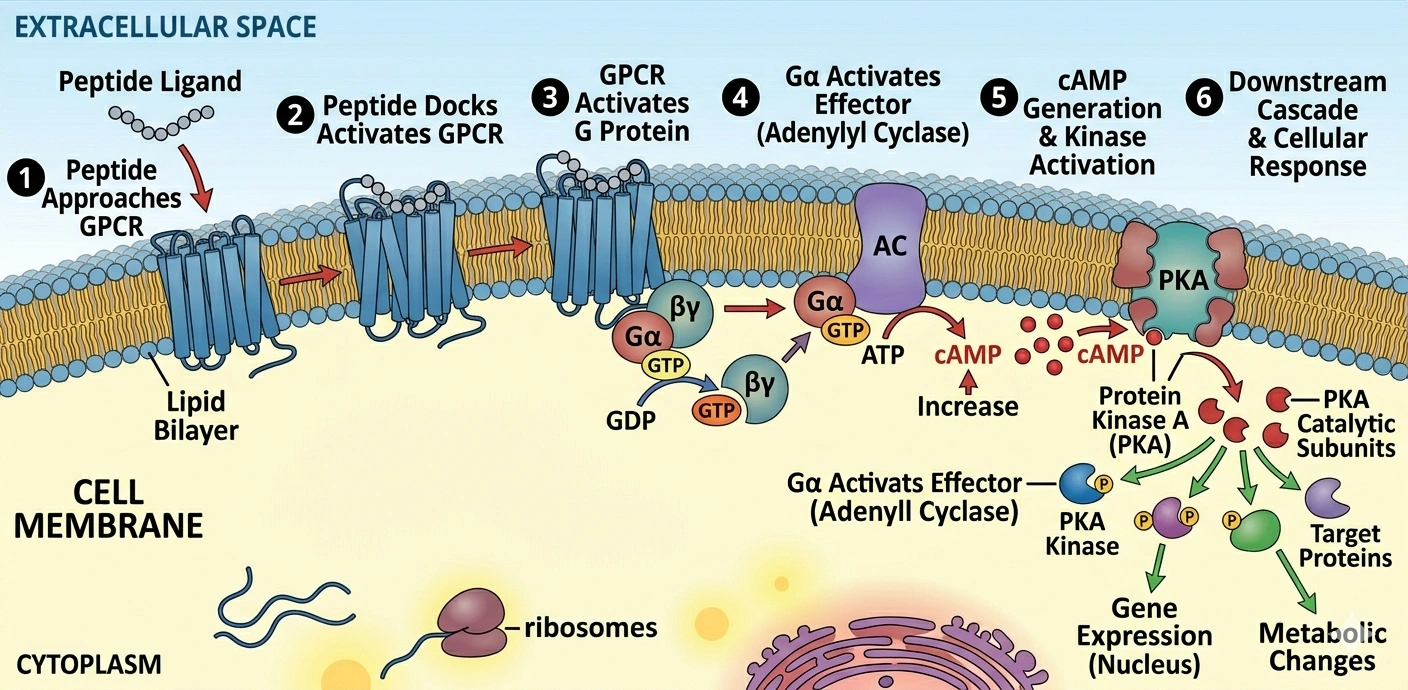
Most peptide receptors are G protein-coupled receptors, or GPCRs. They're found across nearly every tissue in the body. When a peptide binds, the GPCR activates an attached G protein, which kicks off an intracellular cascade: second messengers like cyclic AMP appear, calcium levels shift, kinases switch on, gene expression or protein production changes. The whole sequence runs in milliseconds. One molecule, one receptor, a downstream chain of cellular events.
GPCRs run almost every major biological system: growth hormone release, immune activation, appetite, pain, tissue repair. Therapeutic peptides reach those systems by sending the right message to the right receptor.
For the major peptide categories and which receptors they hit, see the guide to types of peptides.
Why synthetic peptides work like natural ones
A receptor can't tell a synthetic peptide from a natural one if they share the same sequence and the same binding surface. It checks structure, not where the molecule came from.
BPC-157 is a synthetic version of a fragment from a protein found in gastric juice. It shares the amino acid sequence that lets it bind to receptors involved in tissue repair and angiogenesis. The cell gets the same signal it would from the naturally occurring version.
Semaglutide is a modified version of glucagon-like peptide-1, which your gut naturally produces after eating. It binds the same GLP-1 receptor and produces the same appetite-suppressing, insulin-stimulating response. The modification adds a fatty acid chain, which slows enzymatic breakdown and stretches the half-life from minutes to days. The binding surface wasn't touched. The message is the same.
Some therapeutic peptides are sequence-identical to natural ones. Others are analogs, similar enough to hit the right receptor, modified enough to last longer or be more selective. The mechanism is the same either way.
The delivery problem
Most practical questions about peptides come back to this.
Peptides are amino acid chains held together by peptide bonds. Your digestive system is designed to break those bonds. Stomach acid attacks them first, then intestinal proteases, including pepsin and trypsin, chop them into individual amino acids or fragments. Those get absorbed as nutrients. They never reach their intended receptors.
Oral bioavailability for most therapeutic peptides is under two percent. Swallowing a peptide that needs to act systemically is close to not taking it.
Pharmaceutical developers have been trying to solve this for decades — it's not a gap in understanding, it's a hard structural problem.
Topical peptides
Topical peptides skip the bioavailability issue because they don't need to enter the bloodstream. Skincare peptides like GHK-Cu are applied to skin because their target receptors are in the skin itself, in dermal fibroblasts and nearby cells.
Size still matters. Peptides under roughly 500 daltons can penetrate the outer skin barrier through lipid channels between cells. Larger ones stay on the surface without penetration enhancers or liposomes to carry them through. Most signal peptides in skincare are formulated specifically to cross this barrier.
Injectable peptides
Injection sidesteps the gut. A subcutaneous injection deposits the peptide into tissue, where it absorbs into the bloodstream without encountering digestive enzymes. Bioavailability is near-complete. The dose is predictable, and the peptide reaches circulation intact.
Most peptides used in clinical and therapeutic settings are injectable for this reason. For anything requiring systemic action, including growth hormone release, body-wide tissue repair, and immune modulation, injection and oral aren't interchangeable alternatives. They produce different results.
The peptide injections guide covers technique, site selection, and rotation.
Oral peptides: the exceptions
A handful work orally. Desmopressin has a functional oral form. Cyclosporine, used in transplant medicine, achieves meaningful oral absorption. In December 2025, the FDA approved a 25 mg oral semaglutide for weight management, after years of formulation work to get it through the digestion barrier.
What makes these possible: cyclization (connecting the two ends of the peptide chain to block enzymatic attack), D-amino acid substitution (mirror-image amino acids that proteases can't cleave), and lipidation (fatty acid chains that improve membrane permeability). These are engineering choices, not inherent properties of peptides.
BPC-157 comes up a lot in discussions of oral dosing. There's a genuine debate about whether low oral doses do something through local gut receptor activity. For systemic action, though, the bioavailability numbers don't support it yet.
What happens after the peptide binds
Binding starts a cascade. The peptide sends its signal at the membrane; the cell takes it from there.
For GPCR-mediated signaling: peptide binds, receptor activates a G protein, G protein produces second messengers (cyclic AMP is one), second messengers activate kinases, kinases phosphorylate proteins, proteins change gene transcription or enzyme activity. What comes out the other end, whether more collagen, a hormone release, a quieted inflammatory signal, or a metabolic shift, depends on which receptor was triggered and where in the body it lives.
The cascade runs fast. Downstream changes start within minutes. But peptides also clear quickly, with half-lives of minutes to hours for most, broken down by enzymes and excreted. That short residence time is part of why peptides have a different safety profile from drugs that accumulate. The signal doesn't outlast the peptide.
The short half-life also shapes how peptides get dosed. One with a two-hour window often needs daily or twice-daily administration to keep signaling going. CJC-1295 with DAC attaches a drug affinity complex that binds albumin in the blood, stretching its half-life from minutes to several days.
For how half-lives and dosing windows work in practice, see how long peptides stay in your system.
Frequently asked questions
Most peptides don't survive digestion. Your gut is designed to break peptide bonds, which converts a therapeutic peptide into basic amino acids before it reaches any target tissue. Oral bioavailability for most peptides is under two percent. The exceptions, oral semaglutide, desmopressin, and cyclosporine, required significant structural modification or specialized formulation to survive the gastrointestinal environment.
Functionally, yes, provided they share the same receptor-binding structure. Receptors don't check whether a molecule was synthesized in a lab or produced in the body. Some therapeutic peptides are identical to natural sequences. Others are analogs with modifications that improve stability or receptor selectivity. Either way, if the binding surface is the right shape, the cell gets the same signal.
Most peptide effects aren't immediately perceptible. You can't feel a GPCR activating. What you might notice are downstream effects over time: faster recovery, sleep quality changes, skin texture shifts after weeks of consistent use. For peptides that affect measurable biomarkers, such as growth hormone, IGF-1, and inflammatory markers, blood work is the most reliable way to confirm the peptide is doing what it's supposed to do.
It depends on what the peptide is doing. One that triggers hormone release can produce effects within hours of administration. One that works by stimulating collagen production or tissue repair may take four to eight weeks before changes become apparent. Effects accumulate with consistent use, and single doses rarely tell you much.
Because they bind to different receptors, which trigger different intracellular cascades in different tissues. A peptide that activates growth hormone secretagogue receptors in the pituitary does something completely different from one that binds melanocortin receptors in skin or GLP-1 receptors in the gut. The amino acid sequence determines receptor specificity. That specificity determines the biological effect. Treating all peptides as a single category because they're all made of amino acids is like treating all keys as identical because they're all made of metal.
Published by the PEPVi editorial team. Read our editorial standards.