
Peptides and proteins are made of the same stuff: chains of amino acids linked together. But they're not the same thing. The differences come down to size, structure, and what each molecule is actually doing in your body.
They're on a spectrum, not in different categories
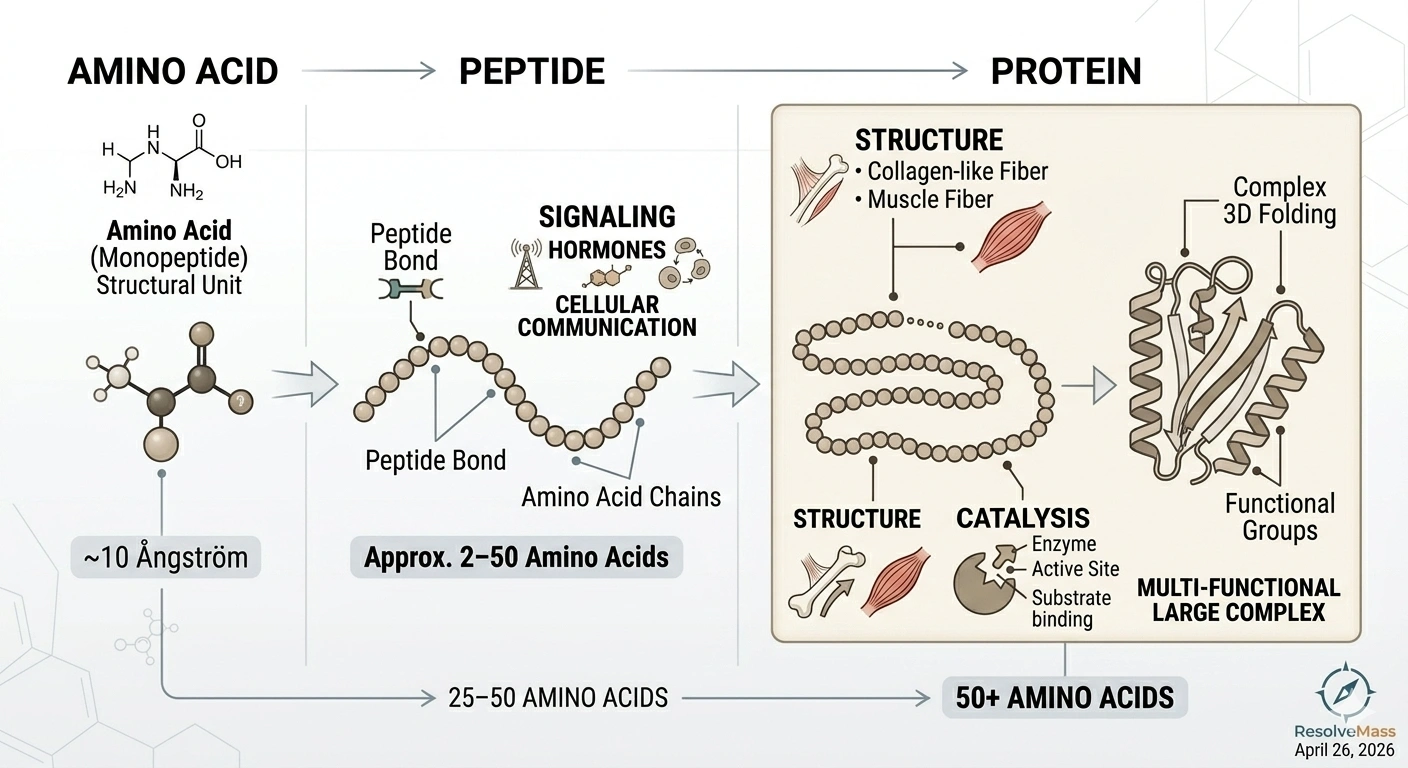
Think of amino acids as building blocks. String a few together and you get a peptide. Keep adding until you have hundreds connected in a chain that folds into a complex 3D shape, and that's a protein.
The dividing line is roughly 50 amino acids. Under that number, the molecule is generally called a peptide. Over it, it's a protein. StatPearls, the NIH's clinical reference, defines peptides as chains of 2 to 50 amino acid residues linked by peptide bonds.
One thing that trips people up: this 50-amino-acid cutoff is a convention, not a hard rule. Insulin has 51 amino acids, which puts it right at the boundary. Most sources classify it as a protein, even though it functions more like a peptide. Britannica describes the distinction as "a little loose," which is a fair characterization.
What matters more than the count is the molecule's structure and what it actually does.
Size determines shape, and shape determines function
A short peptide doesn't have enough chain to fold into a stable 3D structure. Proteins do. That structural complexity is what lets proteins work as enzymes that catalyze chemical reactions, scaffolding materials like collagen and keratin, and antibodies that identify pathogens.
Peptides can't do most of that. But that's not a limitation. It's what makes them useful for something else entirely: signaling.
A peptide doesn't need to be physically large to have biological impact. It needs to be the right shape to fit into a specific receptor on a cell. Once it binds, it triggers a response: hormone release, tissue repair, immune modulation. Oxytocin, vasopressin, and GLP-1 all work this way.
How peptides communicate with cells covers the mechanism in detail. The short version is that size and structure create a functional division: proteins build things; peptides send messages.
Why peptides are more targeted
The signaling role of peptides makes them more specific than most proteins. A large protein has many interaction sites, which creates room for unintended effects. A short peptide has one job: find its receptor, bind, trigger the response.
This specificity is why peptides attract so much interest from pharmaceutical researchers. You can design a short sequence to hit one target with minimal collateral interaction. The tradeoff is stability. Small peptides break down fast in the body, which is why most therapeutic peptides are injected rather than swallowed. What are peptides? covers the delivery challenge in more depth.
You already eat both, and your body converts one into the other
When you eat a chicken breast or a handful of lentils, you're eating protein. Your digestive system breaks that protein into shorter peptide chains, then into individual amino acids, which your body reassembles into whatever it needs. Most of the peptides circulating in your body started out as food protein.
Some foods also contain bioactive peptides that survive digestion in a usable form. Fermented foods like yogurt, kefir, and aged cheese release peptides during fermentation. Milk proteins (casein and whey) yield bioactive peptides during digestion that have documented antihypertensive and immune-modulating effects.
For complete dietary protein, the reliable sources are animal products (meat, fish, eggs, dairy) and plant-based options like legumes, soy, and whole grains. Peptides mostly result from your body processing those proteins rather than something you consume in peptide form directly.
Collagen: where the two concepts collide
Collagen is probably the most instructive example of how peptides and proteins intersect—and why the distinction matters practically.
It's the most abundant protein in the human body, making up a major share of skin, tendons, ligaments, and bone. As a protein, it has a complex triple-helix structure built from three interlocking chains.
The problem with taking collagen orally is that intact collagen molecules are simply too large. Your gut can't absorb them in any meaningful quantity. They get broken down into amino acids just like any other dietary protein, which gives you no specific collagen-synthesis benefit beyond what any protein-rich food would provide. If you're eating collagen-heavy foods like bone broth and expecting a skin benefit, the mechanism doesn't work the way the marketing suggests.
Collagen peptides are different. Through enzymatic hydrolysis, manufacturers pre-digest collagen into short chains of roughly 2 to 20 amino acids. These smaller fragments can be absorbed intact. A systematic review found that collagen peptides reach peak concentration in the bloodstream within about an hour, at which point they can stimulate fibroblasts (the cells that produce new collagen) to increase output.
Another study found that collagen peptides can make up as much as 36% of daily protein intake without disrupting amino acid balance. They don't replace dietary protein. They add a specific functional benefit to a diet that already covers the protein basics.
"Hydrolyzed collagen" and "collagen peptides" on a supplement label mean the same thing. Hydrolysis is the process; peptides are the result. The original whole-protein form, unless it's been hydrolyzed, has very poor oral absorption — research puts bioavailability of intact collagen at around 10–15% versus ~90% for hydrolyzed forms.
If you want to see how collagen peptides fit among other peptide types, the guide to types of peptides covers the broader picture.
Frequently Asked Questions
Technically, yes. But the size difference creates a real functional difference. Peptides (under roughly 50 amino acids) are too short to fold into stable 3D structures, so they can't work as enzymes or structural materials the way proteins do. They function primarily as signaling molecules: binding to receptors and triggering cellular responses. Calling a peptide a "small protein" gets the chemistry right but misses how it actually behaves in the body.
No. Dietary protein provides the raw materials your body uses to build tissues, enzymes, and immune cells. Peptide supplements serve a different purpose: targeted signaling or delivering specific amino acids. Collagen peptides, for instance, don't provide a complete amino acid profile. They work as an addition to a protein-sufficient diet, not a substitute for one.
Intact collagen molecules are too large to cross the gut wall in useful quantities. Collagen peptides have already been enzymatically broken into short chains (2-20 amino acids) that can actually be absorbed. Research suggests bioavailability of hydrolyzed collagen approaches 90%, compared to around 10-15% for intact collagen. If a supplement label says "collagen protein" without specifying hydrolyzed or peptide form, absorption is likely poor.
No. The roughly 50 amino acid boundary is a convention, not a precise rule. Insulin has 51 amino acids and is classified as a protein, even though its function looks more like a peptide. Some sources put the cutoff at 100 amino acids. The categories are useful shorthand, but what matters in practice is the molecule's structure and biological role, not its exact chain length.
Amino acids are individual molecules, the raw units. When two or more link together, you have a peptide. When a peptide chain grows long enough to fold into a stable structure (roughly 50+ amino acids), it becomes a protein. The hierarchy is: amino acids, then peptides, then proteins. Each step adds structural complexity and different biological capabilities.
Published by the PEPVi editorial team. Read our editorial standards.