
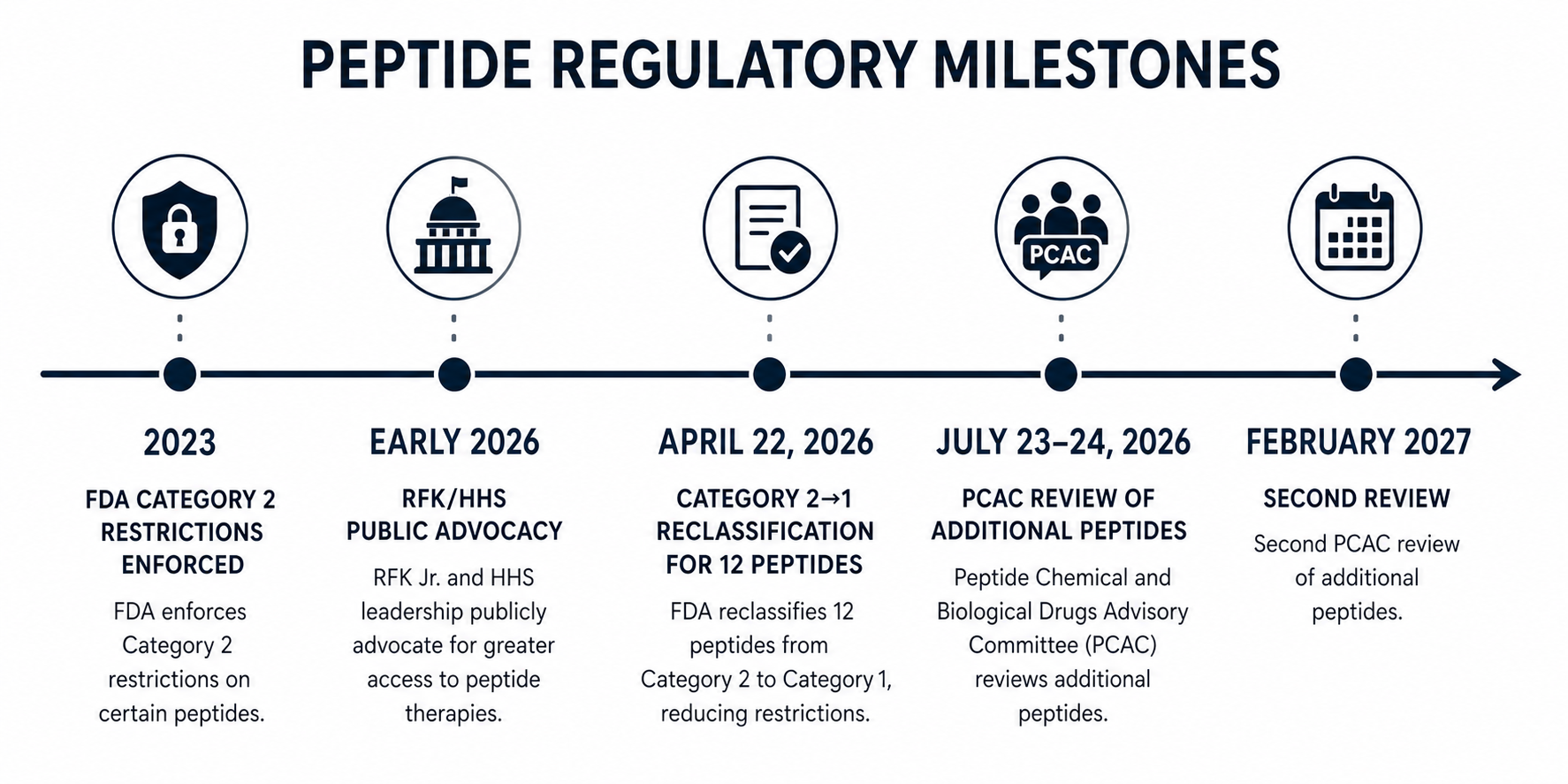
In April 2026, the FDA moved 12 peptides off the list of substances banned from compounding. If you've been following the peptide space, or if you're just now hearing about it, you've probably seen headlines calling this "RFK legalizing peptides." That framing isn't quite right, and understanding why matters more than the headline.
Here's what actually changed, what didn't, and what it means if you're considering peptides for the first time.
The Category System, Explained
The FDA maintains lists of bulk drug substances that licensed compounding pharmacies are either allowed or not allowed to use. These lists are the practical gatekeepers for legal access: if a peptide is on the restricted list, the only way to get it is through the gray market, meaning overseas vendors, research chemical suppliers, or domestic operations in a legal no-man's-land.
There are three relevant categories:
Category 1 means the substance has been nominated for FDA evaluation and has enough safety documentation to allow compounding while that review proceeds. A licensed 503A compounding pharmacy can make it with a valid patient-specific prescription.
Category 2 means the FDA has identified significant safety concerns. Pharmacies cannot compound it, period. The FDA actively takes enforcement action (warning letters, injunctions, product seizures) against pharmacies that use Category 2 substances. This is where BPC-157, TB-500, and ten others lived until April 2026.
Category 3 means insufficient documentation for the FDA to evaluate. Also cannot be compounded.
The April 2026 reclassification moved 12 peptides from Category 2 to Category 1. That's a real change. But it's not the same as FDA approval, and that distinction matters enormously.
The 12 Peptides That Changed Status
Effective April 22, 2026, these peptides were removed from Category 2 and can now be compounded by licensed 503A pharmacies:
| Peptide | Primary use claims |
|---|---|
| BPC-157 | Tissue repair, gut healing, injury recovery |
| TB-500 (Thymosin Beta-4 fragment) | Injury recovery, cell migration, soft tissue repair |
| Epitalon | Longevity, sleep regulation, pineal function |
| GHK-Cu (injectable) | Collagen synthesis, skin health, wound healing |
| KPV | Inflammation reduction, wound healing |
| MOTS-c | Metabolic regulation |
| Semax | Cognitive function, neuroprotection |
| LL-37 (Cathelicidin) | Immune function, antimicrobial activity |
| DiHexa | Cognitive enhancement |
| PEG-MGF | Muscle repair and growth |
| Melanotan II | Skin tanning, sexual function |
| DSIP (Delta Sleep-Inducing Peptide) | Sleep quality |
A note on the evidence behind these claims: it ranges from reasonably supported to almost entirely theoretical. BPC-157 has the most extensive preclinical research of any peptide on this list, but extensive animal data and three small human trials aren't the same thing as proven efficacy. TB-500 has gone slightly further, with Phase 2 wound healing trials showing it accelerated repair and was generally well tolerated. Most of the others have far thinner human data.
The peptide column says what these compounds are claimed to do. The FDA's reclassification says nothing about whether they actually do it.
What Reclassification Doesn't Mean
This is the part that gets compressed or omitted in most coverage of this story.
Reclassification is not FDA approval. The two things have almost nothing in common. FDA drug approval requires Phase 3 clinical trials in large human populations demonstrating both safety and efficacy for a specific indication. None of these peptides have gone through that process. The FDA has not evaluated whether they work. It has not certified their safety at typical consumer doses.
What the reclassification changed: licensed compounding pharmacies are no longer prohibited from making them. That's the full scope of the change.
You still need a valid prescription from a licensed practitioner. The pharmacy still has to follow USP sterile compounding standards. The peptide still has to be made from pharmaceutical-grade bulk substance. None of that changed. Those requirements have always applied to anything a 503A pharmacy dispenses. What changed is that you can now legally access this pathway for these particular peptides.
Think of the prior situation like a door that was locked. The reclassification unlocks it. You still have to walk through it correctly: with a doctor, a prescription, and a legitimate pharmacy.
What RFK's Role Actually Was
Robert F. Kennedy Jr. publicly promoted peptides before his appointment as HHS Secretary, including personal use of BPC-157, and has advocated for easing restrictions on compounds he considers part of natural or functional medicine. His position at HHS has given him real influence over FDA priorities.
Whether his advocacy directly caused this reclassification is murkier than the headlines suggest. The FDA had been reviewing the Category 2 list before his tenure, and the regulatory framework for modifying it already existed. His presence accelerated a process that was already underway and created political conditions that made the FDA more willing to act.
The catch is durability. Enforcement discretion is more fragile than formal rulemaking. A formal rule goes through public comment, generates an administrative record, and is hard for a future administration to reverse quickly. Changes driven primarily by political will can be walked back with the same speed. The April 2026 reclassification is real, but it's not as locked in as a formal FDA approval would be.
Timeline of the 2026 peptide reclassification sequence.
What Changes Practically
If you've been thinking about trying peptides, here's what's actually different as of late April 2026.
The biggest change is where you get them. Before this, anyone who wanted BPC-157, TB-500, or anything else on the now-reclassified list had to go through the gray market: overseas peptide suppliers, research chemical companies, or domestic sources operating without FDA oversight. Quality varied enormously. Contamination with bacteria, heavy metals, or incorrect dosing was a documented risk, not a hypothetical one.
Now you can get these peptides from a licensed 503A compounding pharmacy. That means USP Chapter 797 sterile compounding standards, FDA inspections, and raw material from audited bulk drug suppliers. Not the same thing as an anonymous vial shipped from overseas.
There's also a doctor in the loop. Prescriptions create accountability that self-sourcing doesn't. A practitioner who writes you a script has, in principle, reviewed whether it's appropriate for you, screened for interactions, and has a clinical basis for writing it. Even when the underlying evidence is thin, that layer of oversight has real value.
The tradeoff is cost. Licensed compounding pharmacy pricing typically runs $100–400 per month for a single peptide, plus the consultation fee. The gray market was cheaper precisely because it skipped all of this. If cost was the main reason you were buying unregulated, that math hasn't changed.
For a fuller picture of what regulated versus unregulated sourcing means for safety, including documented adverse events from gray-market peptides, see our safety guide.
What's Coming Next
April 2026 was not the last move. The Pharmacy Compounding Advisory Committee (PCAC) meets July 23–24, 2026, to review a second set of peptides: Ipamorelin, CJC-1295, AOD-9604, Selank, Thymosin Alpha-1, and Kisspeptin-10. Another round follows in February 2027.
The July meeting is worth watching closely. Ipamorelin and CJC-1295 are two of the most widely used peptides in longevity and anti-aging medicine, often combined in protocols targeting growth hormone release. If those clear Category 1, legal access opens up for a significantly larger group of people.
The committee can recommend moving peptides from Category 2 to Category 1, keeping them in Category 2, or raising additional concerns. The FDA isn't bound by the committee's recommendations but follows them closely in practice.
Epitalon is one of the more unusual entries on the reclassified list. It's a synthetic tetrapeptide derived from Soviet-era longevity research, with a different mechanism and evidence base than the tissue-repair peptides. Worth understanding on its own terms.
What the research still doesn't tell you
The reclassification is real. Getting pharmacy-grade peptides from a licensed provider is genuinely better than buying from gray-market vendors.
But none of this changes the underlying evidence base. BPC-157 still has three small human trials. Most of the others have less. The evidence that any of them works as advertised, in humans, at consumer doses, for the outcomes people are seeking: it's thin for most use cases and nonexistent for some.
What changed is where you get them and under what oversight. The reclassification says nothing about whether they work or how safe they are at the doses people use. Those are still open scientific questions.
If you're new to this space, the right first step isn't finding the cheapest compounding pharmacy. It's finding a practitioner who takes the evidence seriously, can tell you honestly what's supported and what isn't, and will screen you appropriately before writing a script.
Frequently Asked Questions
No. FDA approval requires Phase 3 clinical trials demonstrating safety and efficacy in humans. None of the 12 reclassified peptides have gone through that process. Category 1 status means a licensed compounding pharmacy can make them with a prescription — not that the FDA has certified they work or declared them safe.
No. Category 1 allows 503A compounding pharmacies to dispense these peptides, but only with a valid prescription from a licensed practitioner. Buying from gray-market vendors (overseas suppliers, research chemical sites, unlicensed domestic sources) remains legally and medically risky. Those products don't carry the quality controls that licensed pharmacies do.
Any peptide not on an approved compounding list remains restricted. The July 23–24, 2026 PCAC meeting will evaluate Ipamorelin, CJC-1295, AOD-9604, Selank, Thymosin Alpha-1, and Kisspeptin-10. Until that meeting and any resulting FDA action, those remain in their current status.
Not exactly. The FDA moved these peptides from Category 2 to Category 1 through its standard regulatory process. RFK Jr.'s public advocacy and HHS position likely accelerated the timeline and shaped the political environment, but the FDA made the decision through its established framework, not through legislation or executive order.
You can, but the reclassification gives you a better option. Gray-market sourcing still carries quality risks (contamination, incorrect dosing, unknown purity) that a licensed 503A pharmacy eliminates. The tradeoff is cost and the requirement to see a prescribing practitioner. Whether that's worth it depends on your situation, but the quality difference is real.
Published by the PEPVi editorial team. Read our editorial standards.